Abstract
Purpose: To determine the effects of midazolam, 30 ng·mL−1, on altered perception, mood, and cognition induced by ketamine.
Methods: After ketamine was administered to achieve target concentrations of 50, 100, or 150 ng·mL−1 in 11 volunteers, perception, mood, and thought process were assessed by a visual analog scale. Mini-Mental State examination (MMSE) assessed cognition. Boluses of midazolam, 30, 14.5, and 12µg·kg−1, were injected every 30 min to maintain the plasma concentration at 30 ng·mL−1, which was reached 30 min after each injection.
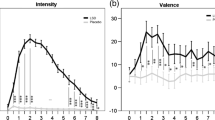
Results: Ketamine produced changes in perception about the body (P<0.01, 0.001, and 0.001 at 30, 60, and 90 min), surroundings (P<0.01 and 0.0001 at 60 and 90 min), time (P<0.002 and 0.0001 at 60 and 90 min), reality (P<0.001 and 0.0001 at 60 and 90 min), sounds (P<0.002 at 90 min), and meaning (P<0.05 at 90 min). Subjects felt less energetic and clearheaded (P<0.02 and 0.05) during ketamine, midazolam, and their co-administration. Ketamine impaired thought process (P<0.003 and 0.0001 and 60 and 90 min). Ketamine and midazolam decreased mean total MMSE and recall scores (P<0.001 for both). Co-administraion reduced the number of subjects with perceptual (body,P<0.01 and 0.001 at 30 and 60 min) and thought process abnormalities. Within the range of observation, co-administration did not affect the changes in mood or recall.
Conclusion: Midazolam attenuates ketamine-induced changes in perception and thought process.
Résumé
Objectif: Déterminer les effets de 30 ng·mL−1 de midazolam sur les changements de perception, d’humeur et de fonction cognitive induits par la kétamine.
Méthode: Après l’administration de kétamine visant à obtenir des concentrations cibles de 50, 100, ou 150 ng·mL−1 chez 11 volontaires, la perception, l’humeur et la fonction cognitive ont été évaluées à l’aide d’une échelle visuelle analogique. L’examen MMS de Folstein et coll. (MMS) a servi à évaluer la fonction cognitive. Des bolus de midazolam de 30, 14,5 et 12µg·kg−1 ont été injectés à toutes les 30 min afin de maintenir la concentration plasmatique à 30 ng·mL−1, concentration atteinte 30 min après chaques injection.
Résultats: La kétamine a modifié la perception du corps (P<0,01; 0,001 et 0,0001 à 30, 60 et 90 min), de l’environnement (P<0,01 et 0,0001 à 60 et 90 min), du temps (P<0,002 et 0,0001 à 60 et 90 min), de la réalité (P<0,001 et 0,0001 à 60 et 90 min), des sons (P <0,002 à 90 min) et du sens (P<0,05 à 90 min). Les sujets se sentaient moins énergiques et moins lucides (P<0,02 et 0,05) pendant l’administration de kétamine, de midazolam et pendant leur co-administration. La kétamine a altéré le processus cognitif (P<0,003 et 0,0001 à 60 et 90 min). La kétamine et le midazolam ont fait baisser les scores totaux moyens de MMS et de mémoire (P<0,001 pour les deux). La co-administration a réduit le nombre de sujets dont les perceptions (corps,P<0,01 et 0,001 à 30 et 60 min) et la fonction cognitive étaient modifiées. Pendant le temps d’observation, la co-administration n’a pas eu d’effet sur les changements d’humeur ou de mémoire.
Conclusion: Le midazolam diminue les changements de perception et de cognition induits par la kétamine.
Article PDF
Similar content being viewed by others
References
Cotman CW, Monaghan DT. Excitatory amino acid neurotransmission: NMDA receptors and Hebb-type synaptic plasticity. Ann Rev Neurosci 1988; 11: 61–80.
Øye I, Paulsen O, Maurset A Effects of ketamine on sensory perception: evidence for a role of N-methyl-D-asparate receptors. J Pharmacol Exp Ther 1992; 260: 1209–13.
Kayama Y, Iwama K. The EEG, evoked potentials, and single unit activity during during ketamine anesthesia in cats. Anesthesiology 1972; 36: 316–28.
Âkeson J, Björkman S, Messeter K, Rosén I. Low-dose midazolam antagonizes cerebral metabolic stimulation by ketamine in the pig. Acta Anaesthesiol Scand 1993; 37: 525–31.
Oguchi K, Arakawa K, Nelson SR, Samson F. The influence of droperidol, diazepam, and physostigmine on ketamine-induced behavior and brain regional glucose utilization in rat. Anesthesiology 1982; 57: 353–8.
White PF, Way WL, Trevor AJ. Ketamine — its pharmacology and therapeutic uses. Anesthesiology 1982; 56: 119–36.
Krystal JH, Karper LP, Seibyl JP, et al. Subanesthetic effects of noncompetitive NMDA antagonist, ketamine, in humans. Psychotomimetic, perceptual, cognitive, and neuroendocrine responses. Arch Gen Psychiatr 1994; 51: 199–214.
Malhotra AK, Pinals DA, Weingartner H, et al. NMDA receptor function and human cognition: the effects of ketamine in healthy volunteers. Neuropsychopharmacology 1996; 14: 301–7.
Bowdle TA, Radant AD, Cowley DS, Kharasch ED, Strassman RJ, Roy-Byrne PP. Psychedelic effects of ketamine in healthy volunteers. Relationship to steady-state plasma concentrations. Anesthesiology 1998; 88: 82–8.
Domino EF, Domino EE, Smith RE, et al. Ketamine kinetics in unmedicated and diazepam-premedicated subjects. Clin Pharmacol Ther 1984; 36: 645–53.
Greenblatt DJ, Ehrenberg BL, Gunderman J, et al. Pharmacokinetic and electroencephalographic study of intravenous diazepam, midazolam, and placebo. Clin Pharmacol Ther 1989; 45: 356–65.
Folstein MF, Folstein SE, McHugh PR “Mini-mental state.” A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975; 12: 189–98.
Chernik DA, Gillings D, Laine H, et al. Validity and reliability of the observer’s assessment of alertness/sedation scale: study with intravenous midazolam. J Clin Psychopharmacol 1990; 10: 244–51.
Stiller RL, Dayton PG, Perel JM, Hug CC Jr. Gas chromatographic analysis of ketamine and norketamine in plasma and urine: nitrogen-sensitive detection. J Chromatogr 1982; 232: 305–14.
Vasiliades J, Owens C. Determination of midazolam in serum by gas chromatography with a nitrogen-sensitive detector. J Chromatogr 1980; 182: 439–44.
Westfall PH, Young SS. Resampling-Based multiple Testing. Examples and Methods forp-Value Adjustment. New York: John Wiley & Sons Inc., 1993: 64–6.
Gilroy J, Meyer JS. Medical Neurology, 3rd ed. New York: MacMillan Publishing Co., Inc., 1979.
Ferrer-Allado T, Brechner VL, Dymond A, Cozen H, Crandall P. Ketamine-induced electroconvulsive phenomena in the human limbic and thalamic regions. Anesthesiology 1973; 38: 333–44.
Carlsson M, Carlsson A. Schizophrenia: a subcortical neurotransmitter imbalance syndrome? Schizophr Bull 1990; 16: 425–32.
Carlsson A, Hansson LO, Waters N, Carlsson ML. Neurotransmitter aberrations in schizophrenia: new perspectives and therapeutic implications. Life Sci 1997; 61: 75–94.
Rosenzweig MR, Leiman AL, Breedlove SM. Biological Psychology. An Introduction to Behavioral, Cognitive and Clinical Neuroscience, 2nd ed. Sunderland: Sinauer Associates, Inc., 1999.
Ghoneim MM, Hinrichs JV, Mewaldt SP, Petersen RC Ketamine; behavioral effects of subanesthetic doses. J Clin Psychopharmacol 1985; 5: 70–7.
Barbee JG. Memory, benzodiazepines, and anxiety: integration of theoretical and clinical perspectives. J Clin Psychiatr 1993; 54: 86–97.
Krystal JH, Karper LP, Bennett A, et al. Interactive effects of subanesthetic ketamine and subhypnotic lorazepam in humans. Psychopharmacology 1998; 135: 213–29.
Gitlin MJ. Clinical manifestations of psychiatric disorders.In: Kaplan HI, Sadock BJ (Eds.). Comprehensive Textbook of Psychiatry, 6th ed. Baltimore: Williams & Wilkins., 1995; 637–69.
Yudofsky SC, Silver JM, Hales RE. Treatment of agitation and aggressive disorders.In: Schatzberg AF, Nemeroff CB (Eds.). Textbook of Psychopharmacology, 1st ed. Washington, D.C.: American Psychiatric Press, Inc., 1995: 735–51.
Krystal JH, D’Souza DC, Karper LP, et al. Interactive effects of subanesthetic ketamine and haloperidol in healthy humans. Psychopharmacology 1999; 145: 193–204.
Murugaiah KD, Hemmings HC Jr. Effects of intravenous general anesthetics of [3H]GABA release from rat cortical synaptosomes. Anesthesiology 1998; 89: 919–28.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Suzuki, M., Tsueda, K., Lansing, P.S. et al. Midazolam attenuates ketamine-induced abnormal perception and thought process but not mood changes. Can J Anaesth 47, 866–874 (2000). https://doi.org/10.1007/BF03019666
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03019666