Abstract
Purpose
Infants with cyanotic congenital heart disease are at risk for cerebral arterial gas embolism (CAGE) from iv infusion lines. Concern about the hazards and difficulty of caring for such patients inside a hyperbaric chamber may deter referral. We report a complex case in which a small infant was managed successfully using a modified hyperbaric oxygen treatment (HBOT) schedule.
Clinical features
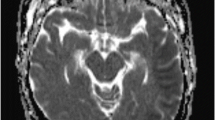
A four-month-old 6.19 kg male infant with a recent Glenn shunt for double-outlet right ventricle had a seizure and became unstable immediately after an iv drug infusion. The patient was sedated, intubated and ventilated and dobutamine was commenced. A computerized tomography (CT) scan performed ten hours later demonstrated three intracranial air bubbles. About ten hours later the patient was referred for HBOT which commenced soon afterwards in a multiplace chamber. Since the rightto-left shunt would greatly increase the risk of decompression illness from breathing hyperbaric air HBOT was modified by the use of an abbreviated schedule at reduced pressure. Two 90-min HBOT sessions were administered within 24 hr at 38 feet of seawater pressure, equivalent to 2.15 atmospheres absolute without any air break. During treatment the infant was ventilated using an Oxford Penlon ventilator. A subsequent CT scan demonstrated the absence of air. After extubation he appeared neurologically intact except for some weakness of the left arm.
Conclusion
Hyperbaric oxygen may be utilized to treat CAGE in small infants with right-to-left shunt and should be commenced promptly.
Objectif
Les enfants atteints de cardiopathie cyanogène congénitale risquent de subir une embolie gazeuse de l’artère cérébrale (EGAC) à partir des cathéters pour perfusion iv. Les préoccupations concernant les dangers et la difficulté de soigner ces patients en chambre hyperbare peuvent retarder les demandes de consultation. Nous présentons un cas complexe de traitement réussi en utilisant un programme modifié d’oxygénothérapie hyperbare (OTHB).
éléments cliniques
Un bébé de quatre mois, de sexe masculin, pesant 6,19 kg, ayant subi récemment une opération de Glenn pour un ventricule droit à double issue, a été victime d’une convulsion et son état est devenu instable immédiatement après la perfusion iv d’un médicament. Nous avons donné un sédatif, intubé et ventilé le patient et amorcé un traitement à la dobutamine. Une tomographie réalisée dix heures plus tard, a montré trois bulles d’air intracrâniennes. Dix heures plus tard environ, le patient a été transféré pour une OTHB, commencée peu après dans une chambre multiplace. Comme le shunt droite-gauche pouvait augmenter beaucoup le risque de maladie de décompression en respirant de l’air hyperbare, l’OTHB a été modifiée par un programme abrégé à pression réduite. Il y a eu deux sessions d’OTHB de 90 min en 24 h à 38 pieds de pression (niveau de la mer), équivalente à 2,15 atmosphères absolues, sans coupure d’air. Pendant le traitement, l’enfant était ventilé avec un appareil Oxford Penlon. Une deuxième tomographie a montré l’absence d’air. Après l’extubation, l’enfant ne présentait aucune déficience neurologique, sauf pour une légère faiblesse du bras gauche.
Conclusion
L’oxygène hyperbare peut être utilisé pour traiter l’EGAC chez de jeunes enfants qui présentent un shunt droite-gauche et devrait être administré sans tarder.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bove AA. Fitness to dive. In: Brubakk AO, Neuman TS (Eds). Bennett and Elliott’s Physiology and Medicine of Diving, 5th ed. Edinburgh: Saunders; 2003: 700–17.
Moon RE, Gorman DF. Treatment of the decompression disorders. In: Brubakk AO, Neuman TS (Eds). Bennett and Elliott’s Physiology and Medicine of Diving, 5th ed. Edinburgh: Saunders; 2003: 600–50.
Messina AG, Leslie J, Gold J, Topkins MJ, Devereux RB. Passage of microbubbles associated with intravenous infusion into the systemic circulation in cyanotic congenital heart disease: documentation by transesophageal echocardiography. Am J Cardiol 1987; 59:1013–4.
Robinson A. Air embolism following CAT scan in a patient with hypoplastic left heart syndrome. Pediatr Cardiol 2003; 24:186.
Heckmann JG, Lang CJ, Kindler K, Huk W, Erbguth FJ, Neundorfer B. Neurologic manifestations of cerebral air embolism as a complication of central venous catheterization. Crit Care Med 2000; 28:1621–5.
Keenan HT, Bratton SL, Norkool DM, Brogan TV, Hampson NB. Delivery of hyperbaric oxygen therapy to critically ill, mechanically ventilated children. J Crit Care 1998; 13:7–12.
Vazquez RL, Spahr RC. Hyperbaric oxygen use in neonates. A report of four patients. Am J Dis Child 1990; 144:1022–4.
Waisman D, Shupak A, Weisz G, Melamed Y. Hyperbaric oxygen therapy in the pediatric patient: the experience of the Israel Naval Medical Institute. Pediatrics 1998; 102:E53.
Joseph MC, McAuley C, Potts MW. A study of the effects of hyperbaric oxygen in infants with cyanotic heart disease. Guys Hosp Rep 1966; 115:73–9.
Hickey PR, Wessel DL. Anesthesia for treatment of congenital heart disease In: Kaplan JA (Ed.). Cardiac Anesthesia, 2nd ed. volume 2. Orlando: Grune & Stratton Inc; 1987: 635–711.
Blanch PB, Desautels DA, Gallagher TJ. Deviations in function of mechanical ventilators during hyperbaric compression. Respir Care 1991; 36:803–14.
Murphy BP, Harford FJ, Cramer FS. Cerebral air embolism resulting from invasive medical procedures. Treatment with hyperbaric oxygen. Ann Surg 1985; 201:242–5.
Blanc P, Boussuges A, Henriette K, Sainty JM, Deleflie M. Iatrogenic cerebral air embolism: importance of an early hyperbaric oxygenation. Intensive Care Med 2002; 28:559–63.
Iwami O, Kawamura J, Hashimoto S, Nakamura M, Suenaga T. Cerebral air embolism (Letter). Am J Neuroradiol 1990; 11:1166.
Voorhies RM, Fraser RA. Cerebral air embolism occurring at angiography and diagnosed by computerized tomography. Case report. J Neurosurg 1984; 60:177–8.
Daneman A, Abou-Reslan W, Jarrin J, Traubici J, Hellmann J. Sonographic appearance of cerebral vascular air embolism in neonates: report of two cases. Can Assoc Radiol J 2003; 54:114–7.
Levy I, Mosseri R, Garty B. Peripheral intravenous infusion - another cause of air embolism. Acta Paediatr 1996; 85:385–6.
Willis J, Duncan C, Gottschalk S. Paraplegia due to peripheral venous air embolus in a neonate: a case report. Pediatrics 1981; 67:472–3.
Neuman TS. Arterial gas embolism and pulmonary barotrauma. In: Brubakk AO, Neuman TS (Eds). Bennett and Elliott’s Physiology and Medicine of Diving, 5th ed. Edinburgh: Saunders; 2003: 557–77.
Walker R. Pulmonary barotrauma. In: Edmonds C, Lowry C, Pennefather J, Walker R (Eds). Diving and Subaquatic Medicine, 4th ed. London: Arnold; 2002: 55–71.
Pennefather J. Physics and physiology. In: Edmonds C, Lowry C, Pennefather J, Walker R (Eds). Diving and Subaquatic Medicine, 4th ed. London: Arnold; 2002: 11–22.
Parbrook GD, Davis PD, Parbrook EO. Diffusion and osmosis. In: Parbrook GD, Davis PD, Parbrook EO (Eds). Basic Physics and Measurement in Anaesthesia, 3rd ed. Oxford: Butterworth Heinemann; 1994: 88–101.
Hart GB, Strauss MB, Lennon PA. The treatment of decompression sickness and air embolism in a monoplace chamber. J Hyperb Med 1986; 1:1–7.
Lavon H, Shupak A, Tal D, et al. Performance of infusion pumps during hyperbaric conditions. Anesthesiology 2002; 96:849–54.
Nuthall G, Seear M, Lepawsky M, Wensley D, Skippen P, Hukin J. Hyperbaric oxygen therapy for cerebral palsy: two complications of treatment. Pediatrics 2000; 106. URL available at http://pediatrics.org/cgi/content/full/106/6/e80.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
LeDez, K.M., Zbitnew, G. Hyperbaric treatment of cerebral air embolism in an infant with cyanotic congenital heart disease. Can J Anesth 52, 403–408 (2005). https://doi.org/10.1007/BF03016284
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03016284