Abstract
Purpose
The objective of this study was to assess the effect of tranexamic acid (TA), a synthetic antifibrinolytic, on blood loss and the need for transfusion of blood products following repeat cardiac valve surgery.
Methods
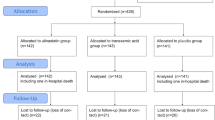
After ethics committee approval, 41 patients scheduled for reoperative valve replacement were enrolled in this randomized, double blind, placebo controlled study. Patients were randomized to receive TA (10 g in 500 ml NSaline) or placebo (NSaline) as an iv bolus over 30 min. after anaesthesia induction and prior to skin incision. Intraoperative blood loss was assessed by estimating blood volume on drapes, weighing surgical sponges, and measuring suction bottle returns. Postoperative blood loss was measured from mediastmal chest tube drainage following surgery. Blood products were transfused according to a standardized protocol.
Results
Patient demographics were similar for age, sex, cardiopulmonary bypass pump time, cross clamp time, surgical time, preoperative haemoglobin, coagulation profile, and the number of valves replaced during surgery. Tranexamic acid administration reduced intraoperative blood loss [median (range)] from 1656 (575–6270) to 720 ml (355–5616) (P < 0.01) and postoperative blood loss from I 170 (180–4025) to 538 ml (135–1465) (Intent to Treat n=41, P < 0.01). The total red blood cells transfused (median, range) was reduced from 1500 (0–9300) ml to 480 (0–2850) ml (P < 0.01) in the TA group. In hospital complications and mortality rates were not reduced in the TA group.
Conclusion
Tranexamic acid reduced blood loss and the need for blood product transfusion and appears to be an effective treatment for patients undergoing reoperative cardiac valvular surgery.
Résumé
Objectif
Évaluer l’influence de l’acide tranéxamique (AT), un antifibrinolytique synthétique, sur les pertes sanguines et le besoin de transfuser des produits sanguins après une réopération cardiaque valvulaire.
Méthodes
Après l’approbation du comité d’éthique, 41 patients ont été recrutés dans cette étude aléatoire, en double aveugle et contrôlée par placebo. Les patients ont été repartis aléatoirement pour recevoir de l’AT (10 g dans 500 ml de sol.phys.) ou un placebo (sol. phys.) en bolusiv en 30 min après l’induction de l’anesthésie et avant l’incision de la peau. Les pertes sanguines peropératoires ont été évaluées en estimant le volume sanguin perdu dans les champs opératoires, en pesant les compresses, et en mesurant le volume sanguin recueilli par aspiration. À la période postopératoire, le sang recueilli par les drains médiastinaux a été mesuré. Les produits du sang ont été administrés selon un protocole standardisé.
Résultats
Les données démographtques étaient identiques pour l’âge, le sexe, la durée de circulation extra-corporelle, du clampage et de la chirurgie, l’hémoglobine préopératoire, le bilan de la coagulation et le nombre de valves remplacées. L’administration d’AT a réduit les pertes sanguines peropératoires [médiane (écart)] de 1665 (572-6 270) à 720 ml (335-5 616) (P < 0,01) et les pertes sanguines postopératoires de 1170 (180-4 025) à 538 ml (135-1 465) (Intention de traitement n=41, P < 0,01). Le nombre total des transfusions d’hématies (médiane, écart) a diminué de 1500 (0-9 300) ml à 480 (0-2 850) ml (P < 0,01) dans le groupe AT. Le taux de complications et de mortalité n’a pas diminué dans le groupe AT durant l’hospitalisation.
Conclusion
L’acide tranéxamique diminue les pertes sanguines et le besoin de transfuser des produits sanguins et semble efficace pour les réopérations en chirurgie cardiaque valvulaire.
Article PDF
Similar content being viewed by others
References
Harker LA, Malpass TW, Branson HE, Hessel EA Slichter SJ. Mechanism of abnormal bleeding in patients undergoing cardiopulmonary bypass: acquired transient platelet dysfunction associated with selective — granule release. Blood 1980; 56: 824–34.
Royston D, Bidstrup BP, Taylor KM, Sapsford RN. Effect of aprotinin on need for blood transfusion after repeat open-heart surgery. Lancet 1987; II: 1289–91.
Lemmer JH Jr, Stanford W, Bonney SL, et al. Aprotinin for coronary bypass operations: efficacy, safety, and influence on early saphenous graft patency. A multicenter, randomized, double-blind, placebo-controlled study. J Thoracic Cardiovasc Surg 1994; 107: 543–53.
Biagini A, Comite C, Russo V, et al. High dose aprotinin to reduce blood loss in patients undergoing redo open heart surgery. Acta Anaesthesial Belg 1992; 43: 181–6.
Cosgrove DM III, Heric B, Lytle BW, et al. Aprotinin therapy for reoperative myocardial revascularization: a placebo-controlled study. Ann Thorac Surg 1992; 54: 1031–8.
Minami K, Notohamiprodjo G, Buschler H, Prohaska W, Reichelt W, Korfer R. Alpha-2 plasmin inhibitorplasmin complex and postoperative blood loss: double-blind study with aprotinin in reoperation for myocardial revascularization (Letter). J Thorac Cardiovasc Surg 1993; 106: 934–5.
Verstraete M. Clinical application of inhibitors of fibrinolysis. Drugs 1985; 29: 236–61.
Royston D. Blood-sparing drugs: aprotinin,tranexamic acid, and ξ-amoncaproic acid. Int Anesthesiol Clin 1995; 33: 155–79.
Horrow JC, Hlavacek J, Strong MD, et al. Prophylactic tranexamic acid decreases bleeding after cardiac operations. J Thorac Cardiovasc Surg 1990; 99: 70–4.
Karski JM, Teasdale SJ, Norman P, et al. Prevention of bleeding after cardiopulmonary bypass with high-dose tranexamic acid. Double-blind, randomized clinical trial. J Thorac Cardiovasc Surg 1995; 110: 835–42.
Horrow JC, Van Riper DF, Strong MD, Brodsky I, Parmet JL. Hemostatic effects of tranexamic acid and desmopressin during cardiac surgery. Circulation 1991; 84: 2063–70.
Yau TM, Carson S, Weisel RD, et al. The effect of warm heart surgery on postoperative bleeding. J Thorac Cardiovasc Surg 1992; 103: 1155–63.
Rousou JA, Engelman RM, Flack JE III, Deaton DW, Owen SG. Tranexamic acid significantly reduces blood loss associated with coronary revascularization. Ann Thorac Surg 1995; 59: 671–5.
Speekenbrink RGH, Vonk ABA, Wildevuur CRH, Eijsman L. Hemostatic efficacy of dipyridamole, tranexamic acid, and aprotinin in coronary bypass grafting. Ann Thorac Surg 1995; 59: 438–42.
Coffey A, Pittmam J, Halbrook H, Fehrenbacher J, Beckman D, Hormuth D. The use of tranexamic acid to reduce postoperative bleeding following cardiac surgery: a double-blind randomized trial. Am Surg 1995; 61: 566–8.
Fremes SE, Wong BI, Lee E, et al. Metaanalysis of prophylactic drug treatment in the prevention of postoperative bleeding. Ann Thorac Surg 1994; 58: 1580–8.
Øvrum E, Holen EA, Abdelnoor M, Øystese R, Ringdal ML. Tranexamic acid (Cyklokapron) is not necessary to reduce blood loss after coronary artery bypass operations. J Thorac Cardiovasc Surg 1993; 105: 78–83.
Bracey AW, Radovancevic R, Radovancevic O, McAllistair HA Jr, Vaughn WK, Cooley DA. Blood use in patients undergoing repeat coronary artery bypass graft procedures: multivariate anaylsis. Transfusion 1995; 35: 850–4.
Bidstrup BP, Royston D, Sapsford RN, Taylor KM. Reduction in blood loss and blood use after cardiopulmonary bypass with high dose aprotinin (Trasylol). J Thorac Cardiovasc Surg 1989; 97: 364–72.
DelRossi AJ, Cernaianu AC, Botros S, Lemole GM, Moore R. Prophylactic treatment of postperfusion bleeding using EACA. Chest 1989; 96: 27–30.
Salzman EW, Weinstein MJ, Weintraub RM, et al. Treatment with desmopressin acetate to reduce blood loss after cardiac surgery. A double-blind randomized trial. N Engl J Med 1986; 314: 1402–6.
Havel M, Teufelshauer H, Knöhl P, etal. Effect of intraoperative aprotinin administration on postoperative bleeding in patients underoing cardiopulmonary bypass operation. J Thorac Cardiovasc Surg 1991; 101: 968–72.
Hardy J-F, Bélisle S. Natural and synthetic antifibrinolytics in adult cardiac surgery: efficacy, effectiveness and efficiency. Can J Anaesth 1994; 41: 1104–12.
Hardy J-F, Desroches J. Natural and synthetic antifibrinolytics in cardiac surgery. Can J Anaesth 1992; 39: 353–65.
Bloom AL. Activation of coagulation and fibrinogen loss after using extracorporeal circulation. J Clin Pathol 1963: 16; 558–61.
Umlas J. Fibrinolysis and disseminated intravascular coagulation in open heart surgery. Transfusion 1976; 16: 460–3.
Ekert H, Montgomery D, Aberdeen E. Fibrinolysis during extracorporeal circulation. Comparison of the effects of disc and membrane oxygenators. Circ Res 1971; 28: 512–7.
Phillips LL, Malm JR, Deterling RA. Coagulation defects following extracorporeal circulation. Ann Surg 1963; 157: 317–26.
Kucuk O, Kwaan HC, Fredrickson J, Wade L, Green D. Increased fibrinolytic activity in patients undergoing cardiopulmonary bypass operation. Am J Hematol 1986; 23: 223–9.
Harker LA. Bleeding after cardiopulmonary bypass (Editorial). N Engl J Med 1986; 314: 1446–8.
Soslau G, Horrow J, Brodsky I. The effect of tranexamic acid on platelet ADP during extracorporeal circulation. Am J Hematol 1991; 38: 113–9.
Horrow JC, Van Riper DF, Strong MD, Grunewald KE, Parmet JL. The dose-response relationship of tranexamic acid. Anesthesiology 1995; 82: 383–92.
Bennett S. Electrocardiographic ST segment changes associated with aprotinin and reversed with heparin in two patients having coronary artery reoperetions. J Cardiothorac Vasc Anesth 1994; 8: 202–5.
Umbrain V, Cristiaens F, Camu F. Intraoperative coronary thrombosis: can aprotinin and protamine be incriminated?. J Cardiothorac Vasc Anesth 1994; 8: 198–201.
Emdadi A, Jamieson WRE, O’Connor JP, et al. Experience with aprotinin in reoperative cardiac surgery. (Abstract) Can J Cardiol. 1993; 9E: 118E.
van der Meer J, Hillege HL, Ascoop CAPL, et al. Aprotinin in aortocoronary bypass surgery: Increased risk of vein-graft occlusion and myocardial infarction? Supportive evidence from a retrospective study. Thromb Haemost 1996; 75: 1–3.
Tyers GFO, Jamieson WRE, Munro AI, et al. Reoperation in biological and mechanical valve populations: fate of the reoperative patient. Ann Thorac Surg 1995; 60: S464–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dryden, P.J., O’Connor, J.P., Jamieson, W.R.E. et al. Tranexamic acid reduces blood loss and transfusion in reoperative cardiac surgery. Can J Anaesth 44, 934–941 (1997). https://doi.org/10.1007/BF03011964
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03011964