Abstract
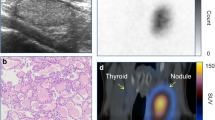
This study was done retrospectively to analyze the ultrasonographic (US) findings in thyroid scintigraphic hot areas (HA). Three-thousand, eight-hundred and thirty-nine consecutive patients who underwent99mTc-pertechnetate (n=3435) or123I (n=457) scintigraphy were analyzed. HA were regarded as present when the tracer concentration was greater than the remaining thyroid tissue, or when hemilobar uptake was observed. High-resolution US examinations were performed with a real-time electronic linear scanner with a 7.5 or 10 MHz transducer. One hundred and four (2.7%) were found to be scintigraphic HA (n=120). US revealed a nodular lesion or welldemarcated thyroid tissue corresponding to the HA in 94 areas (78.4%, Category 1), an ill-defined region with different echogenicity in 13 areas (10.8%, Category 2), and no correlating lesion in 13 areas (10.8%, Category 3). These 104 patients included 43 with adenomatous goiter (59 areas), 33 with adenoma, 11 with Hashimoto’s thyroiditis, 5 with primary thyroid cancer, 4 with euthyroid ophthalmic Graves’ disease (EOG), 3 with hemilobar atrophy or hypogenesis, 2 with hemilobar agenesis, 2 with hypothyroidism with blocking-type TSH-receptor antibodies (TSHRAb), 1 with acute suppurative thyroiditis. Among the 59 adenomatous nodules and 33 adenomas, 51 (86.4%) and 32 (97.0%), respectively, belonged to Category 1. A solitary toxic nodule was significantly larger and occurs more often in older patients than in younger patients. On the other hand, all 17 patients with known autoimmune thyroid diseases including Hashimoto’s thyroiditis, EOG and hypothyroidism with blocking TSHRAb belonged to Category 2 or 3. Possible underlying mechanisms are 1) hyperfunctioning tumors or nodules, 2) localized functioning thyroid tissue freed from autoimmune destruction, inflammation or tumor invasion, 3) congenital abnormality, 4) clusters of hyperactive follicular cells caused by long-term TSH and/or TSHRAb stimulation, 5) asymmetry, etc. Scintigraphic HA are observed in patients with various thyroid diseases and high-resolution US appears to be helpful clinically for the differential diagnosis of the above mentioned disorders.
Similar content being viewed by others
References
Solbiati L, Volterrani L, Riazzatto G, Bazzocchi M, Busilacci P, Candiani F, et al. The thyroid gland with low uptake lesions: evaluation by ultrasound.Radiology 1985; 155: 187–191.
Laszlo H, Steen K. Ultrasonography in the evaluation of cold thyroid nodules.Eur J Endocrinol 1998; 138: 30–31.
Kasagi K, Konishi J, Iida Y, Tokuda Y, Arai K, Endo K, et al. A sensitive and practical assay for thyroid-stimulating antibodies using FRTL-5 thyroid cells.Acta Endocrinol (Copenh) 1987; 115: 30–36.
Konishi J, Iida Y, Kasagi K, Misaki T, Nakashima T, Endo K, et al. Primary myxedema with thyrotropin-binding inhibitor immunoglobulins. Clinical and laboratory findings in 15 patients.Ann Intern Med 1985; 103: 26–31.
Scheible W, Leopold GR, Woo VL, Gosink BB. Highresolution real time ultrasonography of thyroid nodules.Radiology 1979; 133: 413–417.
Torizuka T, Kasagi K, Hatabu H, Misaki T, Iida Y, Konishi J, et al. Clinical diagnostic potential of thyroid ultrasonography and scintigraphy: An evaluation.endocr J 1993; 40: 329–336.
Kasagi K, Hidaka A, Misaki T, Miyamoto S, Takeuchi R, Sakahara H, et al. Scintigraphic findings of the thyroid in euthyroid ophthalmic Graves’ disease.J Nucl Med 1994; 35: 811–817.
Hay DI, Morris CJ. Toxic adenoma and toxic multinodular goiter. In: Braverman EL, Utiger DR (eds),The Thyroid seventh edition. Philadelphia; Lippincott-Raven Publishers, 1996: 566–572.
Miller M, Hamburger IJ. The thyroid scintigram.Radiology 1964; 84: 66–74.
Kusić Z, Becker DV, Saenger EL, Paras P, Gartside P, Wessler T, et al. Comparison of technetium-99m and iodine-123 imaging of thyroid nodules: correlation with pathologic findings.J Nucl Med 1990; 31: 393–399.
Ross SD. Evaluation of the thyroid nodule.J Nucl Med 1991; 32: 2181–2192.
Studer H, Peter JH, Gerber H. Toxic nodular goiter.Clin Endocrinol Metab 1985; 14: 351–372.
Parma J, Duprez L, Van Sande J, Hermans J, Rocmans P, Van Vliet G, et al. Diversity and prevalence of somatic mutations in the thyrotropin receptor and Gs alpha genes as a cause of toxic thyroid adenoma.J Clin Endocrinol Metab 1997; 82: 2695–2701.
Morris JC. Activating mutations of the thyrotropin receptor: unanswered questions.J Clin Endocrinol Metab 1996; 81: 2021–2022.
Hamburger JI. Evolution of toxicity in solitary nontoxic autonomously functioning thyroid nodules.J Clin Endocrinol Metab 1980; 50: 1089–1093.
Branson CJ, Talbot CH, Henry L, Elemenoglou J. Solitary toxic adenoma of the thyroid gland.Br J Surg 1979; 66: 590–595.
Fisher DA, Oddie TH, Johnson DE, Nelson JC. The diagnosis of Hashimoto’s thyroiditis.J Clin Endocrinol Metab 1975; 40: 795–801.
Sulimani RA, Desouki ME. Hashimoto’s thyroiditis presenting as hot and cold nodules.Clin Nucl Med 1990; 15: 315–316.
Yarman S, Mudun A, Alagol F, Tanakol R, Azizlerli H, Oguz H, et al. Scintigraphic varieties in Hashimoto’s thyroiditis and comparison with ultrasonography.Nucl Med Commun 1997; 18: 951–956.
Hayashi N, Tamaki N, Konishi J, Yonekura Y, Senda M, Kasagi K, et al. Sonography of Hashimoto’s thyroiditis.J Clin Ultrasound 1986; 14: 123–126.
Ghose MK, Genuth SM, Abellera RM, Friedman S, Lidsky I. Functioning primary thyroid carcinoma and metastases producing hyperthyroidism.J Clin Endocrinol Metab 1971; 33: 639–646.
Michigishi T, Mizukami Y, Shuke N, Satake R, Noguchi M, Aburano T, et al. An autonomously functioning thyroid carcinoma associated with euthyroid Graves’ disease.J Nucl Med 1992; 33: 2024–2026.
Rossa DG, Testa A, Maurizi M, Satta AM, Aimoni C, Artuso A, et al. Thyroid carcinoma mimicking a toxic adenoma.Eur J Nucl Med 1990; 17: 179–184.
Elizabeth D, Rosen BI, Jerald B, Jaccqueline J, Kirsh CJ. Management of the hot thyroid nodule.Am J Surg 1995; 170: 481–483.
Kasagi K, Hatabu H, Miyamoto S, Takeuchi R, Misaki T, Sakahara H, et al. Scintigraphic findings of the thyroid in hypothyroid patients with blocking-type TSH-receptor antibodies.Eur J Nucl Med 1994; 21: 962–967.
Miyamoto S, Kasagi K, Alam MS, Misaki T, Iida Y, Konishi J. Assessment of thyroid growth stimulating activity of immunoglobulins from patients with autoimmune thyroid diseases by cytokinesis arrest assay.Eur J Endocrinol 1997; 136: 499–507.
Iwata M, Kasagi K, Misaki T, Fujita T, Iida Y, Konishi J. Thyroid hemilobar atrophy in patients diagnosed as having Hashimoto’s thyroiditis.Thyroid 2001; 11: 293–294.
Mikosch P, Gallowitsch HJ, Kresnik E, Molnar M, Gomez I, Lind P. Thyroid hemiagenesis in an endemic goiter area diagnosed by ultrasonography: report of sixteen patients.Thyroid 1999; 9: 1075–1084.
Melnick JC, Stemkowski PE. Thyroid hemiagenesis (Hockey Stick Sign): A review of the world literature and a report of four cases.J Clin Endocrinol Metab 1981; 52: 247–251.
Iida Y, Kasagi K, Misaki T, Arai K, Tokuda Y, Konishi J. Visualization of suppressed normal tissue by Tl-201 in patients with toxic nodular goiter.Clin Nucl Med 1988; 13: 283–285.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iwata, M., Kasagi, K., Hatabu, H. et al. Causes of appearance of scintigraphic hot areas on thyroid scintigraphy analyzed with clinical features and comparative ultrasonographic findings. Ann Nucl Med 16, 279–287 (2002). https://doi.org/10.1007/BF03000108
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03000108