Abstract
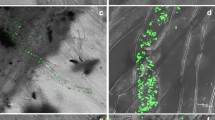
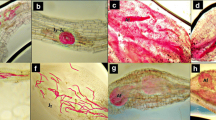
Five accessions ofArabidopsis thaliana, Ws-3, Nd-1, Ler, Col-5 and Oy-0, were inoculated withPeronospora parasitica isolate Emoy2 and the accumulation of camalexin within infected tissues was measured. The variations in camalexin accumulation in various accessions ofA. thaliana induced by a specific (P. parasitica isolate Emoy2) and a non-specific elicitor (UV-B irradiation) were investigated. Phenotypic examination of Emoy2/Oy-0 revealed that susceptibility was characterized by extensive asexual sporulation of the pathogen, whereas early restriction of the pathogen in infected plant tissues, accompanied by chlorosis and necrosis — which are associated with the hypersensitive response — was observed in Nd-1, Ws-3 and Ler. Partial resistance detected in Col-0 was characterized by low to medium sporulation of the pathogen. Camalexin was monitored by high pressure liquid chromatography (HPLC) and found to accumulate during both compatible and incompatible interactions and also following treatment with the abiotic inducer, UV-B. Among the accessions tested, Ws-3 yielded significantly more camalexin than the other accessions, regardless of which inducers (biotic or abiotic) were used. There was no significant correlation between resistance and camalexin accumulation in theA. thaliana/P. parasitica interaction. The results suggest that there is genetic variation in camalexin biosynthesis among accessions ofA. thaliana, rather than variation in types of induction.
Similar content being viewed by others
References
Aarts, N., Metz, M., Holub, E., Staskawicz, B.J., Daniels, M.J. and Parker, J. (1998) Different requirements forEDS1 andNDR1 by disease resistance genes define at least twoR gene-mediated signalling pathways inArabidopsis.Proc. Natl. Acad. Sci. U.S.A. 96:10307–10311.
Allen, R.H.E. and Friend, J. (1983) Resistance of potato tubers to infection byPhytophthora infestans: a structural study of haustorial encasement.Physiol. Plant Pathol. 22:285–292.
Bailey, J.A., Rowell, P.M. and Arnold, G.M. (1980) The temporal relationship between infected cell death, phytoalexin accumulation and the inhibition of hyphal development during resistance ofPhaseolus vulgaris toColletotrichum lindemuthianum.Physiol. Plant Pathol. 17:329–339.
Beggs, C. and Wellman, E. (1985) Analysis of light-controlled anthocyanin formation in coleoptiles ofZea mays L.: The role of UV-B, blue, red and far-red light.Photochem. Photobiol. 41:481–486.
Bittner-Eddy, P., Can, C., Gunn, N., Pinel, M., Tör, M., Crute, I.et al. (1999) Genetic and physical mapping of theRPP13 locus inArabidopsis responsible for specific recognition of severalPeronospora parasitica (downy mildew) isolates.Mol. Plant Microbe Int. 12:792–802.
Botella, M.A., Parker, J.E., Frost, L.N., Bittner-Eddy, P.D., Beynon, J.L., Daniels, M.J.et al. (1998) Three genes ofArabidopsis RPP1 complex resistance locus recognise distinctPeronospora parasitica avirulence determinants.Plant Cell 10:1847–1860.
Conn, K.L., Tewari, J.P. and Dahiya, J.S. (1988) Resistance toAlternaria brassicae and phytoalexinelicitation in rapeseed and other crucifers.Plant Sci. 56:21–25.
Dixon, R.A. and Lamb, C.J. (1990) Molecular communication in interactions between plants and microbial pathogens.Annu. Rev. Plant Physiol. 41:339–367.
Glazebrook, J. and Ausubel, F.M. (1994) Isolation of phytoalexin-deficient mutants ofArabidopsis thaliana and characterization of their interactions with bacterial pathogens.Proc. Natl. Acad. Sci. U.S.A. 91:8955–8959.
Glazebrook, J., Zook, M., Mert, F., Kagan, I., Rogers, E.E., Crute, I.R.et al. (1997) Phytoalexin-deficient mutants ofArabidopsis reveal thatPAD4 encodes a regulatory factor and that fourPAD genes contribute to downy mildew resistance.Genetics 146:381–392.
Hahn, M.C., Bonhoff, A. and Grisebach, H. (1985) Quantitative localisation of the phytoalexin glyceollin I in relation to fungal hyphae in soybean roots infected withPhytophthora megasperma f.sp.glycinea.Plant Physiol. 77:591–601.
Holub, E.B. (1997) Organisation of resistance genes inArabidopsis.in: Crute, I.R., Holub, E.B. and Burdon, J.J. [Eds.] The Gene-for-Gene Relationship in Plant-Parasite Interactions. CAB International, Wallingford, UK. pp. 5–26.
Holub, E.B. and Beynon, J.L. (1997) Symbiology of mouse-ear cress (Arabidopsis thaliana) and Oomycetes.Adv. Bot. Res. 24:227–272.
Knott, D.R. (1989) The wheat rusts — breeding for resistance.in: Frankel, R., Grossman, M., Linskens, H.F., Maliga, P. and Riley, R. [Eds.] Monographs on Theoretical and Applied Genetics. Vol. 12. Springer-Verlag, Berlin, Germany. pp. 170–171.
Kuć, J. (1995) Phytoalexins, stress metabolism, and disease resistance in plants.Annu. Rev. Phytopathol. 33:275–297.
Long, M., Barton-Willis, P., Staskawicz, B.J., Dahlbeck, D. and Keen, N.T. (1985) Further studies on the relationship between glyceollin accumulation and the resistance of soybean leaves toPseudomonas syringae pv.glycinea.Phytopathology 75:235–239.
Mansfield, J.W. (1999) Antimicrobial compounds and resistance: the role of phytoalexins and phytoanticipins.in: Slusarenko, A.J., Fraser, R.S.S. and van Loon, L.C. [Eds.] Mechanisms of Resistance to Plant Diseases. Kluwer Academic Publishers, Amsterdam, the Netherlands. pp. 325–370.
Mansfield, J.W., Bennett, M.W., Bestwick, C.S. and Woods-Tör, A.M. (1997) Phenotypic expression of gene-for-gene interaction involving fungal and bacterial pathogens: variation from recognition to response.in: Crute, I.R., Holub, E.B. and Burdon, J.J. [Eds.] The Gene-for-Gene Relationship in Plant-Parasite Interactions. CAB International, Wallingford, UK. pp. 265–291.
Mayama, S. and Tani, T. (1982) Microspectrometric analysis of the location of avenalumin accumulation in oat leaves in response to fungal infection.Physiol. Plant Pathol. 21:141–150.
Osbourn, A. (1996) Pre-formed anti-microbial compounds and plant defence against fungal attack.Plant Cell 8:1821–1831.
Pedras, M.S.C. and Seguin-Swartz, G. (1992) The blackleg fungus: phytotoxins and phytoalexins.Can. J. Plant Pathol. 14:67–75.
Peuppke, S.G. and VanEtten, H.D. (1976) The relationship between pisatin and the development ofAphanomyces euteiches in diseasedPisum sativum.Phytopathology 66:1174–1185.
Robberecht, R. and Caldwell, M.M. (1978) Leaf epidermal transmittance of ultraviolet radiation and its implication for plant sensitivity to ultraviolet-radiation induced injury.Oecologia 32:277–287.
Rouxel, T., Kolmann, A., Boulidard, L. and Mithen, R. (1991) Abiotic elicitation of indole phytoalexins and resistance toLeptosphaeria maculans within Brassicaceae.Planta 184:271–278.
Skalamera, D., Jobodh, S. and Heath, M.C. (1997) Callose deposition during the interaction between cowpea (Vigna unguiculata) and the monokaryotic stage of the cowpea rust fungus (Uromyces vignea).New Phytol. 136:511–524.
Stapleton, A.E. (1992) Ultraviolet radiation and plants: burning questions.Plant Cell 4:1353–1358.
Thomma, B.P.H.J., Nelissen, I., Eggermont, K., and Broekaert, W.F. (1999) Deficiency in phytoalexin production causes enhanced susceptibility ofArabidopsis thaliana to the fungusAlternaria brassicicola.Plant J. 19:163–171.
Tör, M., Holub, E.B., Brose, E., Musker, R., Gunn, N., Can, C.et al. (1994) Map position of three loci inArabidopsis thaliana associated with isolate-specific recognition ofPeronospora parasitica (downy mildew).Mol. Plant Microbe Int. 7:214–222.
Tsuji, J., Jackson, E.P., Gage, D.A., Hammerschmidt, R. and Somerville, S.C. (1992) Phytoalexin accumulation inArabidopsis thaliana during the hypersensitive reaction toPseudomonas syringae pv.syringae.Plant Physiol. 98:1304–1309.
Author information
Authors and Affiliations
Corresponding author
Additional information
http://www.phytoparasitica.org posting Dec. 16, 2002.
Rights and permissions
About this article
Cite this article
Mert-Turk, F., Bennett, M.H., Mansfield, J.W. et al. Quantification of camalexin in several accessions ofArabidopsis thaliana following inductions withPeronospora parasitica and UV-B irradiation. Phytoparasitica 31, 81–89 (2003). https://doi.org/10.1007/BF02979770
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02979770