Abstract
Background
Technetium-99m methoxyisobutyl isonitrile (Tc-SESTAMIBI) is a substrate of P-glycoprotein and multidrug-resistance associated protein in drug-resistant cells. To assess the clinical effectiveness of Tc-SESTAMIBI for predicting the chemotherapy response to treatment with anthracyclines and vinca alkaloids, we retrospectively evaluated the relationship between the accumulation of Tc-SESTAMIBI and the tumor response.
Methods
Thirteen patients, including 12 advanced cases and 1 relapsed case, were investigated, all of whom had been treated with anthracyclines or a vinca alkaloid regimen. The accumulation of Tc-SESTAMIBI was compared at 10 min and 2 h after Tc-SESTAMIBI administration. The relationship between the accumulation of Tc-SESTAMIBI and the tumor response following treatment with anthracyclines and vinca alkaloids was assessed.
Results
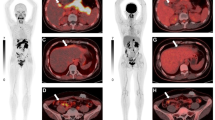
Eight of 13 patients responded to treatment with anthracyclines and vinca alkaloids, whereas 5 patients did not respond to treatment. At 10 min, 6 (75.0%) of the 8 responding patients had a high accumulation of Tc-SESTAMIBI, whereas 4 (80.0%) of the 5 non-responding patients had a low accumulation of Tc-SESTAMIBI. The overall predictive value was 76.9%. The relationship was not statistically significant (Fisher’s test). The difference in the decrease of accumulation of Tc-SESTAMIBI between 10 min and 2 h was not associated with tumor response to treatment in 6 of the responding patients with high accumulation. Two false negative cases and one false positive case were observed, suggesting the presence of another factor contributing to drug sensitivity in tumor response, such as apoptosis-related genes.
Conclusions
Assessment of the initial accumulation of Tc-SESTAMIBI can be a predictive marker of tumor response to treatment with anthracyclines and vinca alkaloids in patients with advanced and relapsed breast cancer. Further studies are required to explore other factors involved in the tumor response to treatment with anthracyclines and vinca alkaloids.
Similar content being viewed by others
Abbreviations
- MDR:
-
Multidrug resistance
- CPA:
-
Cyclophosphamide
- ADM:
-
Adriamycin
- Epi-ADM:
-
Epirubicin
- 5-FU:
-
5-Fluorouracil
References
Gibbons RJ, Verani MS, Behrenbeck T,et al: Feasibility of tomographic Tc-99m-hexakis-2-methoxy-2-methylpropyl-isonitrile imaging for the assessment of myocardial area at risk and the effect of treatment in acute myocardial infarction.Circulation 80:1277–1286, 1989.
Franceschi M, Guimond J, Zimmerman RE,et al: Myocardial clearance of Tc-99m hexakis-2-methoxy-2-methylpropyl isonitrile (MIBI) in patients with coronary artery disease.Clin Nucl Med 15:307–312, 1990.
Savi A, Gerundini P, Zoli P,et al: Biodistribution of Tc-99m methoxy-isobutyl-isonitrile (MIBI) in humans.Eur J Nucl Med 15:597–600, 1989.
Endicott JA, and Ling V: The biochemistry of P-glycoprotein-mediated multidrug resistance.Annu Rev Biochem 58:137–171, 1989.
Roninson IB, ed: Molecular and Cellular Biology of Multidrug Resistance in Tumor Cells. Plenum Publishing Corp, New York: 1991.
Croop JM, Guild BC, Gros P,et al: Genetics of multidrug resistance: relationship of a cloned gene to the complete multidrug resistant phenotype.Cancer Res 47:5982–5988, 1987.
Cole SP, Sparks KE, Fraser K,et al: Pharmacological characterization of multidrug resistant MRP-transfected human tumor cells.Cancer Res 54:5902–5910, 1994.
Piwnica-Worms D, Chiu ML, Budding M,et al: Functional imaging of multidrug-resistant P-glycoprotein with an organotechnetium complex.Cancer Res 53:977–984, 1993.
Hendrikse NH, Franssen EJ, van-der-Graaf WT,et al: 99mTc-sestamibi is a substrate for P-glycoprotein and the multidrug resistance associated protein.Br J Cancer 77:353–358, 1998.
Wosikowski K, Regis JT, Robey RW,et al: Normal p53 status and function despite the development of drug resistance in human breast cancer.Cell Growth Differ 6:1395–1403, 1995.
Wang H, Mohammad RM, Werdell J,et al: p53 and protein kinase C independent induction of growth arrest and apoptosis by bryostain 1 in a highly metastatic mammary epithelial cell line: In vitro versus in vivo activity.Int J Mol Med 1:915–923, 1998.
Orlandi L, Bearzatto A, Abolafio G,et al: Involvement of bcl-2 and p21wafl proteins in response of human breast cancer cell clones to Tomudex.Br J Cancer 81:252–260, 1999.
Sumantran VN, Ealovega MW, Nunez G,et al: Overexpression of bcl-Xs sensitizes MCF-7 cells to chemotherapy-induced apoptosis.Cancer Res 55:2507–2510, 1995.
Sakakura C, Sweeney EA, Shirahama T,et al: Overexpression of bax sensitizes breast cancer MCF-7 cells to cisplatin and etoposide.Surg Today 27:676–679, 1997.
Ohi Y, Kim R, Toge T: Overcoming of multidrug resistance by introducing the apoptosis gene, bcl-Xs, into MRP-overexpressing drug resistant cells.Int J Oncol 16:959–969, 2000.
Duran Cordobes M, Blanchot C, Delmon-Moingeon JC,et al: Tc-99m MIBI uptake by a series of human benign and malignant breast tumor cells: correlation with MDR gene expression. Abstract of the 6th World Congress of Nuclear Medicine and Biology, Sydney, October,Eur J Nucl Med 21 (Suppl):S8, 1994.
Moretti JL, Caglar M, Duran Cordobes M,et al: Can nuclear medicine predict response to chemotherapy?Eur J Nucl Med 22:97–100, 1995.
Kim R, Beck WT: Differences between drug-sensitive and-resistant human leukemic CEM cells in c-jun expression, AP-1 binding activity, and formation of Jun/Fos family dimers, and their association with internucleosomal DNA ladders after treatment with VM-26.Cancer Res 54:4958–4966, 1994.
Gaj CL, Anyanwutaku I, Chang YH,et al: Decreased drug accumulation without increased drug effux in a novel MRP-overexpressing multidrug-resistant cell line.Biochem Pharmacol 55:1199–1211, 1998.
Ford JM and Hait WN: Pharmacology of drugs that alter multidrug resistance in cancer.Pharmacol Rev 42:155–199, 1990.
Boesch D, Gaveriaux C, Jachez B,et al: In vivo circumvention of P-glycoprotein-mediated multidrug resistance of tumor cells with SDZ PSC 833.Cancer Res 51:4226–4233, 1991.
Hyafil F, Vergely C, Du Vignaud P,et al: In vitro and in vivo reversal of multidrug resistance by GF120918, an acridonecarboxamide derivative.Cancer Res 53:4595–4602, 1993.
Pierre A, Leonce S, Perez V,et al: Circumvention of P-glycoprotein-mediated multidrug resistance by S16020-2: kinetics of uptake and efflux in sensitive and resistant cell lines.Cancer Chemother Pharmacol 42:454–460, 1998.
Sikic BI: Modulation of multidrug resistance: at the threshold.J Clin Oncol 11:1629–1635, 1993.
Goldstein LJ: Clinical reversal of drug resistance.Curr Probl Cancer 19:65–124, 1995.
Rischin D, Webster LK, Millward MJ,et al: Cremophor pharmacokinetics in patients receiving 3-, 6-, and 24-hour infusions of paclitaxel.J Natl Cancer Inst 88:97–101, 1996.
Csoka K, Dhar S, Fridborg H,et al: Differential activity of Cremophor EL and paclitaxel in patients’ tumor cells and human carcinoma cell line in vitro.Cancer 79:1225–1233, 1997.
Nabholtz JM, Falkson G, Campos D,et al: A phase III trial comparing doxorubicin (A) and docetaxel (T) (AT) to doxorubicin and cyclophosphamide (AC) as first line chemotherapy for MBC.Proc Am Soc Clin Oncol 18:127a (abstr 485), 1999.
Pluzanska A, Pienkowski T, Jelic S,et al: Phase HI multicenter trial comparing taxol/doxorubicin (AT) vs 5-fluorouracil/doxorubicin and cyclophosphamide (FAC) as a first line treatment for patients with metastatic breast cancer.Breast Cancer Res Treat 57:30 (abstr 21), 1999.
Author information
Authors and Affiliations
Corresponding author
Additional information
Reprint requests to Ryungsa Kim, Department of Surgical Oncology, Research Institute for Radiation Biology and Medicine, Hiroshima University, 1-2-3 Kasumi, Minami-ku, Hiroshima 734-8553, Japan.
About this article
Cite this article
Kim, R., Osaki, A., Hirai, T. et al. Utility of Technetium-99m methoxyisobutyl isonitrile uptake analysis for prediction of the response to chemotherapy in advanced and relapsed breast cancer. Breast Cancer 9, 240–247 (2002). https://doi.org/10.1007/BF02967596
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02967596