Summary
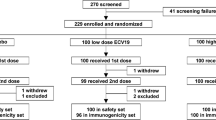
A multicenter, double-blinded, placebo-controlled study was done comparing a 30-μg dose of a single protein recombinant OspA vaccine preparation with a saline placebo for efficacy in prevention of Lyme disease in humans. The OspA vaccine (30-μg dose) or saline placebo was given intramuscularly at day 0,1 month later, and 12 months later. Cases of possible Lyme disease were evaluated clinically and using culture, polymerase chain reaction and immunoblot assays. Safety data are being analyzed separately. 1,634 adult volunteers were enrolled at a single center in New York State. Vaccine efficacy during the first year was 40% and during the second 37%. Compared with placebo, the OspA vaccine significantly reduced the frequency of Lyme disease during the 2-year study period (P<0.04, one-tailed Fisher’s exact test). Vaccine efficacy was restricted to volunteers under 60 years old (50% vs 9%, P<0.03, two-tailed Fisher’s exact test). A recombinant OspA vaccine preparation was found to have moderate activity in preventing Lyme disease in adults under 60 years old from New York State. Reasons for vaccine failure need to be addressed and a risk benefit ratio calculated.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Steere, A. C.: Lyme disease. N. Engl. J. Med. 321 (1989) 586–596.
Fikrig, E., Barthold, S. W., Kantor, F. S., Flavell, R. A.: Protection of mice against the Lyme disease agent by immunizing with recombinant OspA. Science 250 (1990) 553–556.
Fikrig, E., Barthold, S. W., Kantor, F. S., Flavell, R. A.: Long-term protection of mice from Lyme disease by vaccination with OspA. Infect. Immun. 60 (1992) 773–777.
Chang, Y-F., Appel, M. J. G., Jacobson, R. H., Shin, S. J., Harpending, P., Straubinger, R., Patrican, L. A., Mohammed, H., Summers, B. A.: Recombinant OspA protects dogs against infection and disease caused byBorrelia burgdorferi. Infect. Immun. 63 (1995) 3543–3549.
Keller, D., Koster, F. T., Marks, D. H., Hosbach, P., Erdile, L. F., Mays, J. P.: Safety and immunogenicity of a recombinant outer surface protein A Lyme vaccine. JAMA 271 (1994) 1764–1768.
Marks, D. H., Hosbach, P., Koster, F., Wormser, G., Bryant, G., Gwaltney, J., Froeschle, J., Erdile, L., Mays, J.: Clinical studies in humans of outer surface protein A (OspA) vaccine for Lyme disease. In: 6th International Conference on Lyme Borreliosis. Bologna, June 1994, abstr. no. 015.
Schoen, R. T., Meurice, F., Brunet, C. M., Cretella, S., Krause, D. S., Craft, J. E., Fikrig, E.: Safety and immunogenicity of an outer surface protein A vaccine in subjects with Lyme disease. J. Infect. Dis. 172 (1995) 1324–1329.
Sigal, L. H., Adler-Klein, D., Bryant, G., Doherty, T., Haselby, R., Hilton, E., Klempner, M. S., Kunkel, M., Malawista, S.E., Evans, J., Molloy, P., Sabetta, J., Seidner, A., Simon, H. J., Mays, J., Zahradnick, J. M., Marks, D.: Multicenter efficacy trial of a prophylactic recombinantBorrelia burgdorferi (Bd) outer surface protein A (OspA) vaccine for Lyme disease. IDSA 35th Annual Meeting, San Francisco, 13–16 September 1997, abstr. 422.
Centers for Disease Control and Prevention: Case definitions for infectious conditions under public health surveillance. MMWR 46 (1997) RR10: 20–21.
Schwartz I., Wormser, G. P., Schwartz, J. J., Cooper, D., Weissensee, P., Gazumyan, A., Zimmermann, E., Goldberg, N. S., Bittker, S., Campbell, G. L., Pavia, C. S.: Diagnosis of early Lyme disease by polymerase chain reaction amplification or culture of skin biopsics from erythema migrans lesions. J. Clin. Microbiol. 30 (1992) 3082–3088.
Nadelman, R. B., Nowakowski, J., Forseter, G., Goldberg, N. S., Bittker, S., Cooper, D., Aguero-Rosenfeld, M., Wormser, G. P.: The clinical spectrum of early Lyme borreliosis in patients with culture confirmed erythema migrans. Am. J. Med. 100 (1996) 502–508.
Wormser, G. P., Forseter, G., Cooper, D., Nowakowski, J., Nadelman, R. B., Horowitz, H., Schwartz, I., Bowen, S. L., Campbell, G. L., Goldberg, N. S.: Use of a novel technique of cutaneous lavage for diagnosis of Lyme disease associated with erythema migrans. JAMA 268 (1992) 1311–1313.
Nadelman, R. B., Pavia, C. S., Magnarelli, C. A., Wormser, G. P.: Isolation ofBorrelia burgdorferi from the blood of seven patients with Lyme disease. Am. J. Med. 88 (1990) 21–26.
Erdile, L. F., Brandt, M.-A., Warakomski, D. J., Westrack, G. J., Sadziene, A., Barbour, A. G., Mays, J. P.: Role of attached lipid in immunogenicity ofBorrelia burgdorferi OspA. Infect. Immun. 61 (1993) 81–90.
Mathiesen, D. A., Oliver, J. H., Jr.,Kolbert, C. P., Tullson, E. D., Johnson, B. J. B., Campbell, G. L., Mitchell, P. D., Reed, K. D., Telford, S. R. III,Anderson, J. F., Lane, R. J., Persing, D. H.: Genetic heterogeneity ofBorrelia burgdorferi in the United States. J. Infect. Dis. 175 (1997) 98–107.
Fikrig, E., Telford, S. R. III,Wallich, R., Chen, M., Lobet, Y., Matuschka, F. R., Kimsey, R. B., Kantor, F. S., Barthold, S. W., Spielman, A., Flavell, R. A.: Vaccination against Lyme disease caused by diverseBorrelia burgdorferi. J. Exp. Med. 181 (1995) 215–221.
Padilla, M. L., Callister, S. M., Schell, R. F., Bryant, G. L., Jobe, D. A., Lovrich, S. D., DuChateau, B. K., Jensen, J. R.: Characterization of the protective borreliacidal antibody response in humans and hamsters after vaccination withBorrelia burgdorferi outer surface protein A vaccine. J. Infect. Dis. 174 (1996) 739–746.
Wilske, B., Preac Mursic, V., Gobel, U. B., Graf, B., Jauris, S., Soutschek, E., Schwab, E., Zumstein, G.: An OspA serotyping system forBorrelia burgdorferi based on reactivity with monoclonal antibodies and OspA sequence analysis. J. Clin. Microbiol. 31 (1993) 340–350.
ACP Task Force on Adult Immunization and Infectious Diseases Society of America: Guide for adult immunization. 3rd ed. American College of Physicians, Philadelphia 1994, p. 76.
Aguero-Rosenfeld, M. E., Horowitz, H. W., Wormser, G. P., McKenna, D. F., Nowakowski, J., Munoz, J., Dumler, J. S.: Human granulocytic ehrlichiosis (HGE): a case series from a single medical center in New York State. Ann. Intern. Med. 125 (1996) 904–908.
Wormser, G. P., Horowitz, H. W., Dumler, J. S., Schwartz, I., Aguero-Rosenfeld, M.: False-positive Lyme disease serology in human granulocytic ehrlichiosis. Lancet 347 (1996) 981–982.
Wormser, G. P., Horowitz, H. W., Nowakowski, J., McKenna, D., Dumler, J. S., Varde, S., Schwartz, I., Carbonaro, C., Aguero-Rosenfeld, M.: Positive Lyme disease serology in patients with clinical and laboratory evidence of human granulocytic ehrlichiosis. Am. J. Clin. Pathol. 107 (1997) 142–147.
Lovrich, S. D., Callister, S. M., Lim, L. C. L., DuChateau, B. K., Schell, R. F.: Seroprotective groups of Lyme borreliosis spirochetes from North America and Europe. J. Infect. Dis. 170 (1994) 115–121.
Fikrig, E., Barthold, S. W., Persing, D. H., Sun, X., Kantor, F. S., Flavell, R. A.:Borrelia burgdorferi strain 25015: characterization of outer surface protein A and vaccination against infection. J. Immunol. 148 (1992) 2256–2260.
Wormser, G. P.: Prospects for a vaccine to prevent Lyme disease in humans. Clin. Infect. Dis. 21 (1995) 1267–1274.
Wilske, B., Barbour, A. G., Bergstrom, S., Burman, N., Restepo, B. I., Rosa, P. A., Schwan, T., Soutschek, E., Wallich, R.: Antigenic variation and strain heterogeneity inBorrelia spp. Res. Microbiol. 143 (1992) 583–596.
Dykhuizen, D. E., Polin, D. S., Dunn, J. J., Wilske, B., Preac Mursic, V., Dattwyler, R. J., Luft, B. J.:Borrelia burgdorferi is clonal: implications for taxonomy and vaccine development. Proc. Natl. Acad. Sci. USA 90 (1993) 10163–10167.
Milch, L. J., Barbour, A. G.: Analysis of North American and European isolates ofBorrelia burgdorferi with antiserum to a recombinant antigen. J. Infect. Dis. 160 (1989) 351–353.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wormser, G.P., Nowakowski, J., Nadelman, R.B. et al. Efficacy of an OspA vaccine preparation for prevention of lyme disease in New York State. Infection 26, 208–212 (1998). https://doi.org/10.1007/BF02962365
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02962365