Abstract
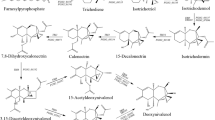
Plants have a high capacity to transform and thereby detoxify deleterious or poisonous compounds, like mycotoxins. The formation of glucose conjugates has a central role in this process. Mammals, however, are able to (partly) release the precursor substances during digestion, reactivating the mycotoxins. This short review provides a brief summary about the metabolism of theFusarium mycotoxins deoxynivalenol and zearalenone in plants. Two examples are discussed in greater detail. First, the formation of deoxynivalenol-3-glucoside in wheat is linked to a quantitative trait locus that is often used forFusarium head blight resistance breeding. Secondly, the metabolism of zearalenone inArabidopsis thaliana results in at least 17 different metabolites, all of which are potentially hazardous for humans and animals.
Similar content being viewed by others
References
Kreuz K, Tommasini R, Martinoia E (1996) Old Enzymes for a New Job: Herbicide Detoxification in Plants. Plant Physiol 111: 349–353
Gareis M, Bauer J, Thiem J, Plank G, Grabley S, Gedek B (1990) Cleavage of zearalenoneglycoside, a “masked” mycotoxin, during digestion in swine. J Vet Med 37: 236–240
Miller JD, Young JC, Trenholm HL (1983) Fusarium toxins in field corn. I. Time course of fungal growth and production of deoxynivalenol and other mycotoxins. Can J Bot 61: 3080–3087
Young CJ, Fulcher GR, Hayhoe JH, Scott PM, Dexter JE (1984) Effect of milling and baking on deoxynivalenol (vomitoxin) content of eastern Canadian wheats. J Agric Food Chem 32 (3): 659–664
Savard ME (1991) Deoxynivalenol fatty acid and glucoside conjugates. J Agric Food Chem 39: 570–574
Sewald N, LepschyvonGleissenthall J, Schuster M, Müller G, Aplin RT (1992) Structure elucidation of a plant metabolite of 4-desoxynivalenol. Tetrahedron 3 (7): 953–960
Poppenberger B, Berthiller F, Lucyshyn D, Sieberer T, Schuhmacher R, Krska R, Kuchler K, Glossl J, Luschnig C, Adam G (2003) Detoxification of theFusarium Mycotoxin Deoxynivalenol by a UDP-glucosyltransferase fromArabidopsis thaliana. J Biol Chem 278 (48): 47905–47914
Berthiller F, Dall’Asta C, Schuhmacher R, Lemmens M, Adam G, Krska R (2005) Masked Mycotoxins: Determination of a Deoxynivalenol Glucoside in Artificially and Naturally Contaminated Wheat by LC-MS/MS. J Agric Food Chem 53: 3421–3425
Engelhardt G, Zill G, Wohner B, Wallnöfer PR (1988) Transformation of theFusarium mycotoxin zearalenone in maize cell suspension cultures. Naturwissenschaften 75: 309–310
Schneweis I, Meyer K, Engelhardt G, Bauer J (2002) Occurrence of zearalenone-4-β-D-glucopyranoside in wheat. J Agric Food Chem 50 (6): 1736–1738
Gareis M (1994) Maskierte Mykotoxine. Übers Tierernährung 22 (1): 104–113
Wallnöfer PR, Preiβ U, Ziegler W, Engelhardt G (1996) Konjugatbildung organischer Schadstoffe in Pflanzen. Z Umweltchem Ökotox 8 (1): 43–46
Engelhardt G, Ruhland M, Wallnöfer PR (1999) Metabolism of mycotoxins in plants. Adv Food Sci 21 (3/4): 71–78
Plasencia J, Mirocha CJ (1991) Isolation and characterization of zearalenone sulfate. Appl Environ Microbiol 57 (1): 146–150
El-Sharkawy S, Selim MI, Afifi MS, Halaweish FT (1991) Microbial transformation of zearalenone to a zearalenone sulfate. Appl Environ Microbiol 57 (2): 549–552
Lemmens M, Scholz U, Berthiller F, Koutnik A, Dall’Asta C, Schuhmacher R, Adam G, Mesterhazy A, Krska R, Buerstmayr H, Ruckenbauer P (2005) A major QTL forFusarium head blight resistance in wheat is correlated with the ability to detoxify the mycotoxin deoxynivalenol. Mol Plant Microbe Interact 18: 1318–1324
Werner U (2006) Characterisation of the effect of theFusarium mycotoxin zearalenone inArabidopsis thaliana. Ph.D. thesis, University of Natural Resources and Applied Life Sciences, Vienna: 1–128
Berthiller F, Werner U, Sulyok M, Krska R, Hauser MT, Schuhmacher R (2006) Liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS) determination of phase II metabolites of the mycotoxin zearalenone in the model plantArabidopsis thaliana. Food Addit Contam 23 (11): 1187–1193
Sulyok M, Berthiller F, Krska R, Schuhmacher R (2006) Development and validation of a liquid chromatography/tandem mass spectrometric method for the determination of 39 mycotoxins in wheat and maize. Rapid Commun Mass Spectrom 20 (18): 2649–2659
Author information
Authors and Affiliations
Corresponding author
Additional information
Financial support: Christian Doppler Society, the Austrian Genome Research Initiative GEN-AU, the Lower Austrian government, the Austrian Science Fund FWF
Rights and permissions
About this article
Cite this article
Berthiller, F., Lemmens, M., Werner, U. et al. Short review: Metabolism of theFusarium mycotoxins deoxynivalenol and zearalenone in plants. Mycotox Res 23, 68–72 (2007). https://doi.org/10.1007/BF02946028
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02946028