Abstract
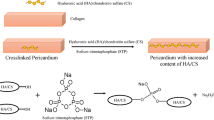
Surface modification of glutaraldehyde fixed bovine pericardium (GFBP) was successfully carried out with hyaluronic acid (HA) derivatives. At first, HA was chemically modified with adipic dihydrazide (ADH) to introduce hydrazide functional group into the carboxyl group of HA backbone. Then, GFBP was surface modified by grafting HA-ADH to the free aldehyde groups on the tissue and the subsequent HA-ADH hydrogel coating. HA-ADH hydrogels could be prepared through selective crosslinking at low pH between hydrazide groups of HA-ADH and crosslinkers containing succinimmidyl moieties with minimized protein denaturation. When HA-ADH hydrogels were prepared at low pH of 4.8 in the presence of erythropoietin (EPO) as a model protein, EPO release was continued up to 85% of total amount of loaded EPO for 4 days. To the contrary, only 30% of EPO was released from HA-ADH hydrogels prepared at pH=7.4, which might be due to the denaturation of EPO during the crosslinking reaction. Because the carboxyl groups on the glucuronic acid residues are recognition sites for HA degradation by hyaluronidase, the HA-ADH hydrogels degraded more slowly than HA hydrogels prepared by the crosslinking reaction of divinyl sulfone with hydroxyl groups of HA. Following a two-week subcutaneous implantation in osteopontin-null mice, clinically significant levels of calcification were observed for the positive controls without any surface modification. However, the calcification of surface modified GFBP with HA-ADH and HA-ADH hydrogels was drastically reduced by more than 85% of the positive controls. The anti-calcification effect of HA surface modification was also confirmed by microscopic analysis of explan ted tissue after staining with Alizarin Red S for calcium, which followed the trend as observed with calcium quantification.
Similar content being viewed by others
References
Ohri, R., S. K. Hahn, P. S. Stayton, A. S. Hoffman, and M. Giachelli (2004) Hyaluronic acid grafting mitigates calcification of glutaraldhyde-fixed bovine pericardium.J. Biomed. Mater. Res. 70A: 159–165.
Golomb, G., F. J. Schoen, M. S. Smith, J. Linden, M. Dixon, and R. J. Levy (1987) The role of glutaraldehyde-induced cross-links in calcification of bovine pericardium used in cardiac valve bioprostheses.Am. J. Pathol. 127: 122–130.
Kim, K. M. (1995) Apoptosis and calcification.Scanning Microscopy 9: 1137–1175.
Schoen, F. J. and R. J. Levy (1999) Tissue heart valves: Current challenges and future research perspectives.J. Biomed. Mater. Res. 47: 439–465.
Vyavahare, N., M. Ogle, F. J. Schoen,et al. (1999) Mechanisms of bioprosthetic heart valve failure: Fatigue causes collagen denaturation and glycosaminoglycan loss.J. Biomed. Mater. Res. 46: 44–50.
Lovekamp, J. and N. Vyavahare (2001) Periodate-mediated glycosaminoglycan stabilization in bioprosthetic heart valves.J. Biomed. Mater. Res. 56: 478–486.
Hunter, G. K., K. S. Wong and J. J. Kim (1988) Binding of calcium to glycosaminoglycans: An equilibrium dialysis study.Arch. Biochem. Biophys. 260: 161–167.
Adrian-Scotto, M., M. Guibbolini, G. Mallet, M. Gaysinski, and D. Vasilescu (2002)23Na NMR study of the interaction between hyaluronan and the bications Ca(++), Mg(++) and Cu(++).J. Biomol. Struct. Dyn. 19: 715–724.
Chang, N. S. and R. J. Boackle (1985) Hyaluronic acid-complement interactions-II. Role of divalent cations and gelatin.Mol. Immunol. 22: 843–848.
Vercruysse, K. P., M. R. Ziebell, and G. D. Prestwich (1999) Control of enzymatic degradation of hyaluronan by divalent cations.Carbohydr. Res. 318: 26–37.
Laurent, T. C. (1998)The Chemistry, Biology and Medical Applications of Hyaluronan and its Derivatives.Wenner-Gren International Series. Vol 72. Portland Press, London, UK.
Fraser, J. R., T. C. Laurent, and U. B. Laurent (1997) Hyaluronan: Its nature, distribution, functions and turnover.J. Intern. Med. 242: 27–33.
Fukuda, K., H. Dan, M. Takayama, F. Kumano, M. Saitoh, and S. Tanaka (1996) Hyaluronic acid increase proteoglycan synthesis in bovine articular cartilage in the presence of interleukin-1.J. Pharmacol. Exp. Ther. 277: 1672–1675.
Goa, K. L. and P. Benfield (1994) Hyaluronic acid. A review of its pharmacology and use as a surgical aid in ophthalmology, and its therapeutic potential in joint discase and wound healing.Drugs 47: 536–566.
Balazs, E. A. and J. L. Delinger (1993) Viscosupplementation: A new concept in the treatment of osteoarthritis.J. Rheumatol. Suppl. 39: 3–9.
Balazs, E. A. (1983) Sodium hyaluronate and viscosurgery. pp. 5–28. In: D. Miller and R. Stegmann (eds.).Healon (Sodium Hyaluronate). A Guide to Its Use in Ophthalmic Surgery. Wiley, NY, USA.
Balazs, E. A. and A. Leshchiner (1986) Cross-linked gels of hyaluronic acid and products containing such gels.US Patent 4,582,865.
Kuo, J. W., D. A. Swann, and G. D. Prestwich (1991) Chemical modification of hyaluronic acid by carbodiimides.Bioconjug. Chem. 2: 232–241.
Illum, L., N. F. Farraj, A. N. Fisher, I. Gill, M. Miglietta, and L. M. Benedetti (1994) Hyaluronic acid ester microspheres as a nasal delivery system.J. Control. Rel. 29: 133–141.
Hahn, K. K. and A. S. Hoffman (2004) Characterization of biocompatible polyelectrolyte complex multiplayer of hyaluronic acid and poly-l-lysin.Biotechnol. Bioprocess Eng. 9: 179–183.
Yeo, Y., N. Bae, and K. Park (2001) Microencapsulation methods for delivery of protein drugs.Biotechnol. Bioprocess Eng. 4: 205–212.
Shu, X. Z., Y. Liu, F. Palumbo, and G. D. Prestwich (2003) Disulfide-crosslinked hyaluronan-gelatin hydrogel films: A covalent mimic of the extracellular matrix forin vitro cell growth.Biomaterials 24: 3825–3834.
Haln, S. K., S. Jelacic, R. V. Maier, P. S. Stayton, and A. S. Hoffman (2004) Anti-inflammatory drug delivery from hyaluronic acid hydrogels.J. Biomat. Sci. Polym. Ed. 15: 1111–1119.
Steitz, S. A., M. Y. Speer, M. D. McKee,et al. (2002) Osteopontin inhibits mineral deposition and promotes regression of ectopic calcification.Am. J. Pathol. 161: 2035–2046.
Pouyani, T. and G. D. Prestwich (1994) Functionalized derivatives of hyaluronic acid oligosaccharides: Drug carriers and novel biomaterials.Bioconjug. Chem. 5: 339–347.
Bitter, T. and H. Muir (1962) A modified uronic acid carbazole reaction.Anal. Biochem. 4: 330–334.
Liaw, L., D. E. Birk, C. B. Ballas, J. S. Whitsitt, J. M. Davidson, and B. L. Hogan (1998) Altered wound heating in mice lacking a functional osteopontin gene (spp1).J. Clin. Invest. 101: 1468–1478.
Bulpitt, P. and D. Aeschlimann (1999) New strategy for chemical modification of hyaluronic acid: Preparation of functionalized derivatives and their use in the formation of novel biocompatible hydrogels.J. Biomed. Mater. Res. 47: 152–169.
Hermanson, G. T. (1996)Bioconjugate Techniques. pp. 121. Academic Press, San Diego, USA.
Giachelli, C. M. and S. Steitz (2000) Osteopontin: A versatile regulator of inflammation and biomineralization.Matr. Biol. 19: 615–622.
Lee, W. K., K. D. Park, D. K. Han, H. Suh, J. C. Park, and Y. H. Kim (2000) Heparinized bovine pericardium as a novel cardiovascular bioprosthesis.Biomaterials 21: 2323–2330.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hahn, S.K., Ohri, R. & Giachelli, C.M. Anti-calcification of bovine pericardium for bioprosthetic heart valves after surface modification with hyaluronic acid derivatives. Biotechnol. Bioprocess Eng. 10, 218–224 (2005). https://doi.org/10.1007/BF02932016
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02932016