Abstract
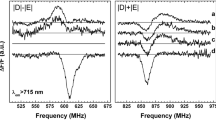
Fluorescence detected magnetic resonance (FDMR) spectra and fluorescence emission spectra at 4.2 K of chlorophyll-proteins isolated and purified from barley thylakoids are presented. The FDMR spectra show the occurrence of chlorophylla triplet states in all five chlorophyll-proteins studied, namely Chl a -P1, Chl a -P2, Chl a -P3, Chl a/b -P1 and Chl a/b -P2.
The presence of more than one chlorophyll triplet each associated with a chlorophyll emitting at a specific wavelength gives rise to a characteristic wavelength dependence of the FDMR spectrum of chlorophyll-proteins. The zero field splitting parameters measured, combined with the observed fluorescence emission wavelengths suggest that three types of interactions of the Mg atom of chlorophylla occur in these proteins: a type similar to that in the parallel dimer (Chla·H2O)2, seen at 721 nm for Chl a -P1 leading to a positive FDMR signal; a type like that in Chla· 2 pyridine also giving a positive FDMR signal, seen in Chl a -P2 and Chl a -P3; and a third type similar to that in Chla· 2H2O leading to a negative FDMR signal, seen for Chl a -P1 at 679 nm, and for Chl a/b -P1 and Chl a/b -P2.
The FDMR spectrum in the antenna of photosystem I (Chl a -P1) can probably be ascribed to that of a trap formed by a pair of interacting chlorophylla molecules, indicating that the organisation of chlorophyll in the antenna may not in principle be very different from that in the photosystem I reaction centre, and that it contains approximately plane-parallel chlorophylla pairs. Chl a -P2 and Chl a -P3 do not show a long wavelength (>700 nm) emission, suggesting a much weaker interaction between chlorophyll molecules in these proteins compared to that in Chl a -P1. For Chl a/b -P1 and Chl a/b -P2 the absence of a long wavelength emission and the observation of zero field splitting (ZFS) parameters similar to that of monomeric Chla·2H2O both indicate the absence of strong interactions between chlorophylla molecules in these proteins also, and it is suggested that chlorophylla and chlorophyllb molecules occur in interacting pairs.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Beck, J., G. H Kaiser, I. U. von Schütz &H. C. Wolf: Optically excited triplet states in the bacteria Rhodopseudomonas sphaeroides «wild type» detected by magnetic resonance in zero field. Biochim. Biophys. Acta 634, 165–173 (1981)
Bent, S. J. van der: Interaction of light and microwaves with photosynthetic pigments. Ph. D. Thesis, Wageningen (1977)
Bent, S. J. van der, P. A. de Jager &T. J. Schaafsma: Optical detection and electronic simulation of magnetic resonance in zero magnetic field of dihydroporphin in free base. Rev. Sci. Instr. 47, 117–212 (1976)
Blankenship, R. E., T. J. Schaafsma &W. W. Parson: Magnetic field effects on radical pair intermediates in bacterial photosynthesis. Biochim. Biophys. Acta 461, 297–305 (1977)
Brakel, G. H. van, T. J. Schaafsma & J. J. S. van Rensen: Fluorescence detected magnetic resonance (FDMR) of whole algae and the Anacystis nidulans CP-I complex. Isr. J. of Chem., in press. (1981)
Clarke, R. H., S. P. Jagannathan &W. R. Leenstra: Optical-microwave double resonance spectroscopy of in vivo chlorophyll. Photochem. Photobiol. 32, 805–808 (1980)
Clarke, R. H., W. R. Leenstra &W. G. Hagar: Observation of a triplet state in chlorophyll protein 668 via optically detected magnetic resonance. FEBS Lett. 99, 207–209 (1979)
Dorp, W. G. van, T. J. Schaafsma, M. Soma &J. H. van der Waals: Investigation of the lowest triplet state of free base porphin by microwave induced changes in its fluorescence. Chem. Phys. Lett., 21, 221–225 (1973)
El-Sayed, M. A.: Phosphorescence-microwave multiple resonance spectroscopy. In: MTP International Review of Science, D. A. Ramsay ed., Butterworths, London, Vol. III pp. 119–153 (1973)
Fuad, N., D. A. Day, S. W. Thorne & I. J. Ryrie: Fluorescence of chlorophyll-protein complexes from higher plants in isolation and upon reconstitution into artificial membranes. Abstracts XIII. Int. Bot. Congress, Sydney, Australia, 1981, p. 223.
Geschwind, S., ed., Electron Paramagnetic Resonance, Plenum, New York, (1972)
Hiller, R. G., B. L. Møller &G. Høyer-Hansen: Characterization of six putative photosystem I mutants in barley. Carlsberg Res. Commun.45, 315–328 (1980)
Hoff, A. J., Govindjee &J. C. Romijn: Electron spin resonance in zero magnetic field of triplet states of chloroplasts and subchloroplast particles. FEBS Lett., 73, 191–196 (1977)
Hoff, A. J., H. Rademaker, R. van Grondelle &L. N. M. Duysens: On the magnetic field dependence of the yield of the triplet state in reaction centers of photosynthetic bacteria. Biochim. Biophys. Acta 460, 547–554 (1977)
Katz, J. J., J. R. Norris &L. L. Shipman: Models for reaction center and antenna chlorophyll. Brookhaven Symp. Biol. 28, 16–55 (1977)
Klimov, V. V., E. Dolan &B. Ke: EPR properties of an intermediary acceptor (pheophytin) in P. S.-II reaction centers at low temperature. FEBS Lett. 112, 97–100 (1980)
Knox R. S. &R. L. van Metter: Fluorescence of light-harvesting chlorophyll a/b-protein complexes: Implications for the photosynthetic unit. In: Chlorophyll organisation and Energy Transfer in Photosynthesis. Ciba Found. Symp. 61, pp. 177–190. Elsevier, Amsterdam (1979)
Kooyman, R. P. H.: Complexes and aggregates of chlorophylls. Ph. D. Thesis Wageningen (1980)
Kooyman, R. P. H., T. J. Schaafsma &J. F. Kleibeuker: Fluorescence spectra and zero field magnetic resonance of chlorophyll-a-water complexes. Photochem. Photobiol. 26, 235–240 (1970)
Machold, O., D. J. Simpson &B. L. Møller: Chlorophyll-proteins of thylakoids from wild type and mutants of barley (Hordeum vulgare L.). Carlsberg Res. Commun. 44, 235–254 (1979)
Markwell, J. P., J. P. Thornber &R. T. Boggs: Higher plant chloroplast: Evidence that all the chlorophyll exists as chlorophyll-protein complexes. Proc. nat. Acad. Sci. USA 76, 1233–1235 (1979)
Mau, A. W. H. &M. Puza: Phosphorescence of chlorophylls. Photochem. Photobiol. 25, 601–603 (1977)
Olson, J. M.: Bacteriochlorophyll-proteins from green bacteria. In: The Photosynthetic Bacteria, R. K. Clayton and W. R. Sistrom eds., Plenum, New York, Chapter 8 (1978)
Schaafsma, T. J.: ODMR of the chlorophylls in vitro and in vivo. In: ODMR Spectroscopy: Techniques and Application to Biophysical Systems, R. H. Clarke ed., Wiley, New York. Chapter 8, in press (1981)
Schaafsma, T. J.: The study of the electronic structure of porphyrin molecules in solid matrices by optical and magnetic resonance spectroscopy. Proc. Fourth Int. Sem. Energy Transfer in Condensed Matter, Prague, June 30–July 3, Czechoslovakia (1981)
Schmidt, J. &J. H. van der Waals: Optical detection of zero field transitions in phosphorescent triplet states. Chem. Phys. Lett. 2, 640–642 (1968)
Searle, G. F. W., J. Barber, L. Harris, G. Porter &C. J. Tredwell: Picosecond laser study of fluorescence life times in spinach chloroplast photosystem I and photosystem II preparations. Biochim. Biophys. Acta 459, 390–401 (1977)
Searle G. F. W., G. H. van Brakel, W. F. J. Vermaas, A. van Hoek & T. J. Schaafsma: Chlorophyll triplets and energy transfer in isolated P.S.-I, chlorophyll proteins from bluegreen algae. Proc. 5th. Int. congress on Photosynthesis, Halkidiki, Greece, in press, (1980)
Searle, G. F. W., R. B. M. Koehorst & T. J. Schaafsma: Fluorescence detected magnetic resonance of isolated chlorophyll-proteins containing chlorophylla andb: energy transfer studies. Proc. Fourth Int. Sem. Energy Transfer in Condensed Matter, Prague, June 30th–July 3rd, Czechoslovakia (1981)
Simpson, D. J. &D. von Wettstein: Macromolecular physiology of plastids. XIV. Viridis mutants in barley: genetic, fluorescopic and ultrastructural characterisations. Carlsberg Res. Commun. 45, 283–314 (1980)
Waals, J. H. van der &M. S. de Groot: Magnetic interactions related to phosphorescence. In: The Triplet State, A. B. Zahlan ed., The University Press, Cambridge, pp. 101–133 (1967)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Searle, G.F.W., Koehorst, R.B.M., Schaafsma, T.J. et al. Fluorescence detected magnetic resonance (FDMR) spectroscopy of chlorophyll-proteins from barley. Carlsberg Res. Commun. 46, 183 (1981). https://doi.org/10.1007/BF02906496
DOI: https://doi.org/10.1007/BF02906496