Summary
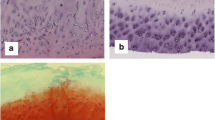
The distribution of the interstitial collagens I, II and III was analyzed immunohistochemically in cartilage and bone samples from 32 patients with degenerative osteoarthrosis at various morphological stages. The alterations observed showed a very patchy, focal distribution demonstrating significant heterogeneity in the tissue reaction. In minor osteoarthrotic lesions a focal pericellular deposition of collagens III and I was seen, while the majority of the interterritorial matrix reacted exclusively with collagen II antibodies. These changes were first seen in the superficial cartilage layer. At the more advanced stages of osteoarthrosis, particularly when osteophytic bone spur formation was present, extensive changes in the expression of collagen types in the pericellular matrix was revealed with extensive and overlapping localization of collagens I, II and III in the whole cartilage. These observations support the suggestion that degenerative cartilage shows a collagen type “switch”. In addition, it was demonstrated that the interterritorial cartilage matrix was still mainly composed of collagen II even in advanced lesions. These observations may explain some of the previous discrepancies reported.
Similar content being viewed by others
References
Abbott J, Holtzer H (1966) The loss of phenotypic traits by differentiated cells. J Cell Biol 28:473–487
Adam M, Deyl Z (1983) Altered expression of collagen phenotype in osteoarthrosis. Clin Chim Acta 133:25–32
Ayad S, Chambers CA, Berry L, Shuttleworth CA, Grant ME (1986) Type VI collagen and glycoprotein MFPI are distinct components of the extracellular matrix. Biochem J 236:299–302
Barsky SH, Rao NC, Restrepo C, Liotta L (1984) Immunocytochemical enhancement of basement membrane antigens in pathology: application in diagnostic pathology. Am J Clin Pathol 82:191–194
Benya PD, Shaffer JD (1982) Dedifferentiated chondrocytes reexpress the differentiated collagen phenotype when cultured in agarose gels. Cell 30:215–224
Cordell JL, Falinin B, Erber WN, Ghosh AH, Abdulaziz Z, MacDonald S, Pulford AF, Stein H, Mason DY (1984) Immunoenzymatic labeling of monoclonal antibodies using immune complexes of alkaline phosphatase and monoclonal anti-alkaline phosphatase. J Histochem Cytochem 32:219–225
Eyre D, McDevitt C, Billingham M, Muir H (1980) Biosynthesis of collagen and other matrix proteins by articular cartilage in experimental osteoarthrosis. Biochem J 188:823–837
Gay S, Müller P, Lemmen C, Remberger K, Matzen K, Kühn K (1976) Immunohistochemical study on collagen in cartilagebone metamorphosis and degenerative osteoarthrosis. Klin Wochenschr 54:969–976
Gay S, Rhodes RK (1981) Immunhistologic demonstration of distinct collagen in normal and osteoarthritic joints. Sem Arthritis Rheum [Suppl 1] 11:43–44
Goldwasser M, Astley T, van der Rest M, Glorieux FH (1982) Analysis of the type of collagen present in ostearthritic human cartilage. Clin Orthop 167:296–302
Hsu SM, Raine L, Fanger H (1981) A comparative study of the peroxidase-antiperoxidase method and an avidin-biotin complex method for studying polypetide hormones with radioimmunoassay antibodies. Am J Clin Pathol 75:734–739
Keene DR, Engvall E, Glanville RW (1988) Ultrastructure of type VI collagen in human skin and cartilage suggests an anchoring function for this filamentous network. J Cell Biol 107:1995–2006
Layman DL, Sokoloff L, Miller EJ (1972) Collagen synthesis by articular chondrocytes in monlayer culture. Exp Cell Res 73:107–112
Lipiello L, Hall D, Mankin HJ (1977) Collagen synthesis in normal and osteoarthritic human cartilage. J Clin Invest 59:593–600
Mankin HJ, Lipiello L (1970) Biochemical and metabolic abnormalities in articular cartilage from osteoarthritic human hips. J Bone Jt Surg 52A: 424–434
Mayne R, Vail MS, Mayne PM, Miller EJ (1976) Changes in type of collagen synthesized as clones of chick chondrocytes grow and eventually lose division capacity. Proc Natl Acad Sci USA 73:1674–1678
Mayne R (1989) Cartilage collagens: What is their function, and are they involved in articular disease? Arthritis Rheum 32:241–246
Nerlich A, Schleicher E (1991) Immunohistochemical localization of extracellular matrix components in human diabetic glomerular lesions. Am J Pathol 139:889–899
Nimni M, Deshmukh K (1973) Differences in collagen metabolism between normal and osteoarthritic human articular cartilage. Science 181:751–752
Poole AR (1986) Changes in the collagen and proteoglycan of articular cartilage in arthritis. Rheumatology 10:316–371
Ronziere MC, Riccard-Blum S, Tiollier J, Hartmann DJ, Garrone R, Herbage D (1990) Comparative analysis of collagens solubilized from human foetal and normal and osteoarthritic adult articular cartilage, with emphasis on type VI collagen. Biochim. Biophys. Acta 1038:222–230
Sandy JD, Adams ME, Billingham ME, Plaas A, Muir H (1984) In vivo and in vitro stimulation of chondrocyte biosynthetic activity in early experimental osteoarthritis. Arthritis Rheum 27:388–397
Timpl R, Wick G, Gay S (1977) Antibodies to distinct types of collagens and procollagens and their application in immunohistology. J Immunol Meth 18:165–175
Venn MF, Maroudas A (1977) Chemical composition and swelling of normal and osteoarthrotic femoral head cartilage. Ann Rheum Dis 36:121–129
von der Mark K, Kirsch T, Aigner T, Reichenberger E, Nerlich A, Weseloh G, Stöß H (1992) The fate of chondrocytes in osteoarthritic cartilage: regeneration, dedifferentiation or hypertrophy. In: Kuettner K, Schleyerbach R, Peyron JG, Hascall VC (eds) Articular cartilage and osteoarthritis. Raven Press, New York, pp 221–234
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nerlich, A.G., Wiest, I. & von der Mark, K. Immunohistochemical analysis of interstitial collagens in cartilage of different stages of osteoarthrosis. Virchows Archiv B Cell Pathol 63, 249–255 (1993). https://doi.org/10.1007/BF02899269
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02899269