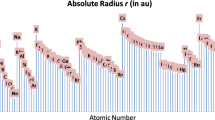
Abstract
The characteristic radii for univalent cations and anions were defined by the classical turning point of the electron movement in an ion. The numerical results of the elements from firstto third-rows in the periodic table were obtained usingab initio method. The results correlate quite well with Pauling ionic radii and Shannon and Prewitt ionic radii.
Similar content being viewed by others
References
Shannon, R. D., Revised effective ionic radii and systematic studies of interatomic distances in Mides and chalcogenides,Acta Cryst., 1976, 1432: 751.
Marcus, Y., Ionic radii in aqueous solutions,Chem. Rev., 1988, 88: 1475.
Pauling, L.,The Nature of the Chemical Bond, 3rd ed, New York: Cornel University Press Ithaca, 1960.
Moller, T.,Inorganic Chemistry (a Modern Introduction), New York: John Willey Sons, 1982, 70.
Mackay, K. M., Mackay, R. A.,Introduction to Modnn Inorganic Chemistry. 3rd ed. London: International Textbook Company, 1981, 26.
Yang, Z. Z., Niu, S. Y., A sort of radius to describe the size of an atom,Chinese Science Bulletin, 1991, 36: 964.
Niu, S. Y., Yang, Z. Z., Atomic boundary radii,Acta Chimica Sinica, 1994, 52:551.
Yang, Z. Z., Davidson, E. R., Evaluation of a characteristic atomic radius by anab initio method,Int J Quantum Chem, 1997,62: 47.
Kohn, W., Sham, L. I., Self-consistent equations including exchange and correlation effects,Phys. Rev., 1965, 140: A1133.
Davidson, E. R.,MELD Program Description in MOTECC, New York: ESCOM, 1990.
Robert, C. W., Melvin, J. A., Willian, H. B.,Handbook of Chemistry and Physics, Florida: CRC Press, INC, 1995.
Author information
Authors and Affiliations
About this article
Cite this article
Yang, Z., Li, G., Zhao, D. et al. Theoretical study on characteristic ionic radii. Chin.Sci.Bull. 43, 1452–1455 (1998). https://doi.org/10.1007/BF02884123
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02884123