Summary
Studies were performed on the clinical basis of chemotherapy for human cancer with uracil and l-(2′-tetrahydrofuryl)-5-fluorouracil (FT).
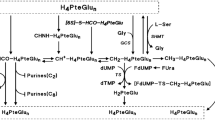
In 62 operated patients with stomach, breast, and uterine cancers, the concentration of 5FU and FT were compared in serum, tumor and normal tissues 1, 2, 4, 6, 8, and 12 hours following the administration of 300 mg of UFT (300 mg of FT plus 672 mg uracil, a uracil/FT molar ratio of 4), or FT alone. It was found that the level of 5-FU in tumor tissue remained above 0.05 mcg/g over 12 hours. This value for 5-FU corresponds to a minimum inhibitory concentration in thein Vitro experiment with L1210 cells. Blood levels of 5-FU increased up to 0.1 mcg/ml 1 hour after UFT administration and then decreased below 0.05 mcg/ml. The drug concentration in normal tissues was lower than that of the tumor tissues.
On the basis of the above findings and phase I study, a protocol of UFT-therapy was made and applied for the treatment of gastric cancer. Our patients were given an oral dose of 300 mg of UFT twice a day per 50 kg body weight (12 mg/kg BW). Therapeutic effects were detectable in 7 of 20 cases. In addition, a combination of mitomycin C (6 mg i.v. weekly) with UFT seemed to improve the response rate (5/7). Diarrhea (15%) and skin pigmentation (10%) were major side effects of UFT.
Similar content being viewed by others
References
Kimura K: Chemotherapy of cancer. Nippon I ji-Shimpo No. 2766: 3–11, 1977 (Jpn)
Fujii S, et al: Effects of coadministration of uracil or cytosine on the antitumor activity of clinical doses of 1-(2-tetrahydrofuryl)-5-fluorouracil (FT) and level of 5-fluorouracil in rodents. Gann 70: 209–214, 1979
Taguchi T, et al: Effect of uracil on the antitumor activity of futraful. Gan to Kagakuryoho 5: 1161–1165, 1978 (Jpn)
Taguchi T, et al: Determination of 5-fluorouracil levels in tumor, blood, and other tissues. Gann to Kagakuryoho 5: 1167–1172, 1978 (Jpn)
Marunaka T, Umeno Y: Quantitative determination of 1,3-bis (tetrahydro-2-furanyl)-5-fluoro-2,4-pyrimidine-dione and its metabolites in plasma by high-pressure liquid chromatography and gas chromatographymass fragmentography. J Chromatg 157: 321–330, 1978
Mukheerjee L, Heidelberger C: Studies on fluorinated pyrimidine. IX The degradation of 5-fluorouracil-6-C14. J Biol Chem 235: 433–437, 1960
Jato J, Windheuser JJ: 5-fluorouracil and derivatives in cancer chemotherapy. III In vitro enhancement of antitumor activity of 5-fluorouracil (FU) and 5-fluoro-2′-deoxyuridine (FUDR). J Pharm Sci 62: 1975–1978, 1973
Jato JG, et al: Effect of deoxyuridine coadministration on toxicity and antitumor activity of fluorouracil and fluorouridine. J Pharm Sci 64: 943–946, 1975
Schumacher HJ, et al: Potentiation of the teratogenic effects of 5-fluorouracil by natural pyrimidines. II Biochemical aspects. Teratology 2: 99–105, 1969
Ikenaka K, et al: Effect of uracil on metabolism of 5-fluorouracil in vitro. Gann 70: 353–359, 1979
Author information
Authors and Affiliations
Additional information
This work was supported in part by the Grant-in-Aid for Cancer Research from the Ministry of Health and Welfare, and the Ministry of Education, Science and Culture.
Rights and permissions
About this article
Cite this article
Kimura, K., Suga, S., Shimaji, T. et al. Clinical basis of chemotherapy for gastric cancer with uracil and 1-(2′-tetrahydrofuryl)-5-fluorouracil. Gastroenterol Jpn 15, 324–329 (1980). https://doi.org/10.1007/BF02774302
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02774302