Abstract
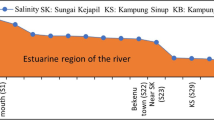
When river water mixes with sea water in estuary area, the concentrations of the dissolved element in river water may be changed by either a simple physical mixing process or some complex chemical processes. It has been clarified in the Chikugogawa River estuary area that the change in concentrations of SO 2−4 , BO 3−3 , Mg2+, Ca2+ and F− is only due to the mixing process but the change in concentrations of SiO 2−3 and Al3+ is due to the chemical process in addition to the mixing process.
Similar content being viewed by others
References
Gast, J. A. andT. G. Thompson (1958): Determination of the alkalinity and borate concentration of sea water. Anal. Chem.,30, 1594.
Bien, G. S., D. E. Contols andW. H. Thomas (1958): The removal of soluble silicate from fresh water entering the Sea. Geochim. Cosmochim. Acta,14, 35.
 ,Sverdrup, H. V.
et al. (1942): The Oceans, their physics, chemistry, and general biology, Prentice-Hall ed. 193 p.
,Sverdrup, H. V.
et al. (1942): The Oceans, their physics, chemistry, and general biology, Prentice-Hall ed. 193 p.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hosokawa, I., Ohshima, F. & Kondo, N. On the Concentrations of the Dissolved Chemical Elements in the Estuary Water of the Chikugogawa River. Journ. Oceanogr. Soc. Japan 26, 1–5 (1970). https://doi.org/10.1007/BF02764549
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02764549



 ,
,