Abstract
Objective
Miltefosine, an alkyl phospholipid has been found effective against visceral leishmaniasis (VL) in adults in various studies. The authors safety, tolerance and efficacy of Miltefosine and compared with available gold standard anti-leishmanial drug, Amphotericin B, a parenteral formulation in children with VL.
Methods
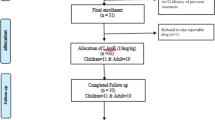
All consecutive children aged 1 yr to 14 yr, presented with fever, splenomegaly and positive LD body in splenic smear examination, admitted in pediatric ward of Nalanda Medical college and Child care center between 1st July 03 to 30th June 05 were taken for study. Patients were randomized into four groups. Group-1 and 2 patients were given Miltefosine in dose of 2.5 mg/Kg day o.d. or b.i.d. per orally to a maxilpum of 100 mg and group 3 and 4 Amphotericin B at a dose of 1 mg/Kg/day (total: 15 mg/Kg). All patients were followed at completion of therapy, 3 months and 6 months for clinical response, splenic size and parasitologically.
Results
Out of 125 children, 44 were in group-1, 20 in group-2, 38 in group-3 and 23 in group 4, 124 patients had parasitological cure with relapse in one patient of group 1 during follow up. One patient in-group II had no response with first course but became parasitologically negative with 2nd course of Miltefosine. In-group I, one patient had persistent splenomegaly and found to have associated portal hypertension. Final cure rate with Miltefosine and Amphotericin B was 93.2%, 95%, 92.1% and 91.3% in-group 1, 2, 3 and 4 respectively, which are statistically insignificant. Majority of patients had pancytopenia. Eievated″. AL T (>3 times of normal) were seen in 28, 11, 19 and 13 patients of group 1, 2, 3 and group 4 respectively which returned to normal in subsequent follow up. Raised BUN was observed more in patients who got Amphotericin B i.e. 65.42% and 73.91% in-group 3 and 4 respectively. GI side effects i.e. diarrhea and vomiting were observed in 26 and 23 patients in-group 1 and 2 respectively.
Conclusion
Miltefosine is safe, well tolerable, and highly effective and has same efficacy as Amphotericin B in newly diagnosed and SAG resistant children with visceral leishmaniasis.
Similar content being viewed by others
References
Desjeux P. Leishmaniasis: public health aspects and control.Clin Dermatol 1996; 14: 417–423.
Croft SL, Neal RA, Pendergast W, Chan JH. The activity of alkylphosphocholines and related derivatives against Leishmania donovani.Biochemical Parasitol 1987; 42: 2633–2636.
Kuhlencord A, Maniera T, Eibl H, Unger C. Hexadecyl phosphocholine: oral treatment of visceral leishmaniasis in mice.Antimicro agents Chenothe 1992; 36: 1630–1634.
Jendrossek V, Handrick R. Membrane Targeted Anticancer Drugs: Potent Inducer of Apoptosis and putative Radiosesitisers.Curr Med Chem Anti-Canc Agents 2003; 3(5): 343–353.
Sundar S, Jha TK, Thakur CP, Engel J, Sindermann H, Fishcer C, Junge K, Bryceson A, Berman J. Oral Miltefosine for Indian Visceral leishmaniasis.N Engl J Med 2002; 347: 1739–1746.
Sundar S, Jha TK, Sindermann H, Junge K, Bachmann P, Berman J. Oral miltefosine in children with mild to moderate Indian visceral leishmanisis.Pediatr Infect Dis J 2003; 22(5): 434–438.
Bryceson A. A policy for leishmaniasis with respect to the prevention and control of drug resistance.Trop Med Int Health 2001; 6: 928–934.
Jha SN, Singh NKP, Jha TK. Changing response to diamidine compounds in cases of kala-azar unresponsive to antimonial.J Assoc Physicians Indian 1991; 39: 314–316.
Thakur CP, Kumar M, Pandey AK. Comparison of regimes of treatment of antimony resistant kala-azar patients: a randomized controlled study.Am J Trop Med Hug 1991; 45: 435–441.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, U.K., Prasad, R., Mishra, O.P. et al. Miltefosine in children with visceral leishmaniasis: A prospective, multicentric, cross-sectional study. Indian J Pediatr 73, 1077–1080 (2006). https://doi.org/10.1007/BF02763048
Issue Date:
DOI: https://doi.org/10.1007/BF02763048