Abstract
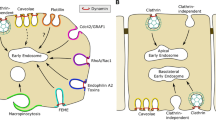
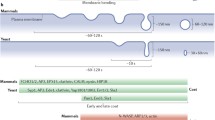
The fluorescence probe [1-(4-trimethylammonium]-6-phenyl-1,3,5-hexatriene (TMA-DPH) displays properties relevant for both monitoring endocytosis kinetics and assessing membrane fluidity by fluorescence-anisotropy measurements (1). Thus, it is, possible with this probe to follow the evolution of membrane fluidity during endocytosis, from the very beginning of the process, i.e., the formation of endocytic vesicles. In most cases, endocytosis is known to start with clathrin-coated vesicles. Still, there are more and more arguments in favor of a complementary endocytic pathway without clathrin. In this article we present membrane-fluidity data for very early endocytosis, which allow an upper limit to be determined for the contribution of a putative nonclathrin pathway. We show that this limit is markedly higher for bone marrow-derived macrophages than for mouse fibroblasts of the L929 cell line.
Similar content being viewed by others
References
Illinger, D., Duportail, G., Mély, Y., Poirel-Morales, N., Gérard, D., and Kuhry, JG. (1995) A comparison of the fluorescence properties of TMA-DPH as a probe for plasma membrane and for endocytic membrane.Biochim. Biophys. Acta. 1239, 58–66.
Robinson, M. S., Watts, C., and Zerial, M. (1996) Membrane dynamics in endocytosis.Cell 84, 13–21.
Watts, C. and Marsh, M. (1992) Endocytosis: what goes in and how?J. Cell Sci. 103, 1–8.
Schmid, S. L. (1997) Clathrin-coated vesicle formation and protein sorting: an integrated process.Ann. Rev. Biochem. 66, 511–548.
Sandvig, K. and van Deurs, B. (1994) Encocytosis without clathrin.Trends Cell Biol. 4, 275–277.
Lamaze, C. and Schmid, S. L. (1995) The emergence of clathrin-independent pinocytic pathways.Curr. Op. Cell Biol. 7, 573–580.
Hansen, S. H., Sandvig, K., and van Deurs, B. (1991) The preendosomal compartment comprises distinct coated and non coated endocytic vesicle populations.J. Cell Biol. 113, 731–741.
Daukas, G. and Zigmond, S. H. (1985) Inhibition of receptor-mediated but not fluid-phase endocytosis in polymorphonuclear leukocytes.J. Cell. Biol. 101, 1673–1679.
Madshus, I. H., Sandvig, K., Olsnes, S., and van Deurs, B. (1987) Effect of reduced endocytosis by hypotonic choc and potassium depletion on the infection of HEp-2 cells by picornavirus.J. Cell Physiol. 131, 14–22.
Cupers, P., Veithen, A., Kiss, A., Baudhuin, P., and Courtoy, P. J. (1994) Clathrin polymerization is not required for bulk-phase endocytosis in rat fetal fibroblasts.J. Cell Biol. 127, 725–735.
Damke, H., Baba, T., van der Blick, A. M., and Schmid, S. L. (1995) Clathrin-independent pinocytosis is induced in cells overexpressing a temperature-sensitive mutant of dynamin.J. Cell Biol. 131, 69–80.
Damke, H. (1996) Dynamin and receptor-mediated endocytosis.FEBS Lett. 389, 48–51.
Hinshaw, J. E. and Schmid, S. L. (1995) Dynamin self-assembles into rings suggesting a mechanism for coated vesicle budding.Nature 374, 190–192.
Illinger, D. and Kuhry, J. G. (1994) The kinetic aspects of intracellular fluorescence labeling with TMA-DPH support the maturation model for endocytosis in L929 cells.J. Cell Biol. 125, 783–794.
Illinger, D., Poindron, P., Glasser, N., Modollel, M., and Kuhry, J. G. (1990) The plasma-membrane internalization and recycling is enhanced in macrophages upon activation with gamma-interferon and lipopolysaccharide, a study susing the fluorescent probe trimethylaminodiphenylhexatriene.Biochim. Biophys. Acta,1030, 82–87.
Kuhry, J. G., Duportail, G., Bronner, C., and Laustriat, G. (1985) Plasma membrane fluidity measurements on whole living cells by fluorescence anisotropy of trimethylamino-diphenylhexatriene.Biochim. Biophys. Acta 845, 60–67.
Liaubet, A., Egret-Charlier, M., and Kuhry, J. G. (1994) Influence of the clathrin coat on the membrane lipidic organization of endocytic vesicles: a fluorescence study.Biochim. Biophys. Acta 1195, 164–168.
Smythe, E., and Warren, G. (1991) The mechanism of receptor-mediated endocytosis.Eur. J. Biochem. 202, 689–699.
Paddenberg, R., Wiegand, C., and Jockush, B. M. (1990) Characterization of the coated vesicle uncoating ATPase: tissue distribution association with and activity on intact coated vesicles.Eur. J. Cell Biol. 52, 60–66.
Belcher, J. D., Hamilton, R. L., Brady, S. E., Hornick, C. A., Haeckle, W. J., and Havel, R. J. (1987) Isolation and characterization of three endosomal fractions from the liver of estradiol-treated rats.Proc. Natl. Acad. Sci. USA 84, 6785–6789.
Nonnenmacher, H., Illinger, D., Kuhry, J. G., Kirn, A., and Braunwald, J. (1994) Contrary results on mouse hepatitis virus type 3 susceptibility in A/J mouse hepatocytes of phosphatidylserine treatment and of a hypercholesterolaemic diet: no correlation with membrane fluidity levels.Biochem. Biophys. Res. Com. 204, 1299–1304.
Lakowicz, J. R. (1983) InPrinciples of Fluorescence Spectroscopy, Plenum, New York, p. 145.
Lentz, B. R. (1988) Membrane fluidity from fluorescence anisotropy measurements, inSpectroscopic Membrane Probe (Loew, L. M., ed.), CRC, Boca Raton, FL, pp. 13–43.
Swanson, J. A., and Watts, C. (1995) Macropinocytosis.Trends Cell Biol. 5, 424–428.
Swanson, J. A. (1989) Phorbol esters stimulate macropinocytosis and solute flow through macrophages.J. Cell Sci. 94 135–142.
Veithen, A., Cupers, P., Baudhuin, P., and Courtoy, P. J. (1996) v-Src induces constitutive macropinocytosis in rat fibroblasts.J. Cell Sci. 109, 2005–2012.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Coupin, G., Kuhry, JG. Differentiation between clathrin-dependent and clathrin-independent endocytosis by means of membrane fluidity measurements. Cell Biochem Biophys 30, 25–34 (1999). https://doi.org/10.1007/BF02737883
Issue Date:
DOI: https://doi.org/10.1007/BF02737883