Abstract
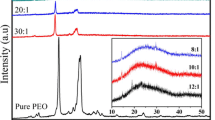
A new solid polymer electrolyte, (PEG)xLiClO4, consisting of poly(ethylene)glycol of molecular weight 2000 and LiClO4 was prepared and characterized using XRD, IR, SEM, DSC, NMR and impedance spectroscopy techniques. XRD and IR results show the formation of the polymer-salt complex. The samples with higher salt concentration are softer, less opaque and less smooth compared to the low salt concentration samples. DSC studies show an increase in the glass transition temperature and a decrease in the degree of crystallinity with increase in the salt concentration. Melting temperature of SPEs is lower than the pure PEG 2000. Room temperature1H and7Li NMR studies were also carried out for the (PEG)xLiClO4 system. The1H linewidth decreases as salt concentration increases in a similar way to the decrease in the crystalline fraction and reaches a minimum at aroundx = 46 and then increases.7Li linewidth was found to decrease first and then to slightly increase after reaching a minimum atx = 46 signifying the highest mobility of Li ions for this composition. Room temperature conductivity first increases with salt concentration and reaches a maximum value (σ = 7.3 × 10−7 S/cm) atx = 46 and subsequently decreases. The temperature dependence of the conductivity can be fitted to the Arrhenius and the VTF equations in different temperature ranges. The ionic conductivity reaches a high value of ∼10 −4S/cm close to the melting temperature.
Similar content being viewed by others
References
Armand M B, Chabagno J M and Duclot M 1978Second international meeting on solid electrolytes (ed.) C A Vincent (Scotland: St. Andrews) p. 20
Berthier C, Gorecki W, Minier M, Armand M B, Chabagno J M and Rigaud P 1983Solid State Ionics 11 91
Binesh N and Bhat S V 1998J. Polym. Sci. Part B: Polym. Phys. 36 1201
Boukamp B A 1989EQUIVCRT program V 3.39 (The Netherlands: University of Twente)
Bruce P G and Vincent C A 1993J. Chem. Soc. Faraday Trans. 89 3187
Cohen M H and Turnbull D 1959J. Chem. Phys. 31 1164
Fenton D E, Parker J M and Wright P V 1973Polymer 14 589
Fulcher G S 1925J. Am. Ceram. Soc. 8 339
Gibbs J H and DiMarzio E A 1958J. Chem. Phys. 28 373
Gray F M 1991Solid polymer electrolyte-fundamentals and technological applications (New York: VCH Publishers, Inc.)
Hikichi K and Furuichi J 1965J. Polym. Sci. A3 3003
Johansson A, Wendsjö A and Tegenfeldt J 1992Electrochim. Acta 37 1487
Kelly I, Owen J R and Steele B C H 1984J. Electroanal. Chem. 168 467
Le Nest J F, Gandini A and Cheradane H 1988Br. Polym. J. 20 253
Li X and Hsu S L 1984J. Polym. Sci., Polym. Phys. Ed. 22 1331
Ratner M A 1987Polymer electrolyte reviews (eds) J R MacCallum and C A Vincent (London: Elsevier Applied Science)Vol. 1, pp 185–195
Ratner M A and Shriver D F 1988Chem. Rev. 88 109
Shi J and Vincent C A 1993Solid State Ionics 60 11
Tamman G and Hesse W 1926Z. Anorg. Allg. Chem. 19 245
Tanzella F L, Baily W, Frydrych D, Farrington G C and Story H S 1981Solid State Ionics 5 681
Vincent C A 1987Prog. Solid State Chem. 17 145
Vogel H 1922Phys. Z. 22 645
Watanabe M, Togo M, Sanui K, Ogata N, Kobayashi T and Ohtaki Z 1984Macromolecules 17 2908
Weston J E and Steele B C H 1982Solid State Ionics 7 81
Whittingham M S, Weppner W and Schulz H (eds) 1988Solid State Ionics 28-30 1
Wright P V 1975Br. Polym. J. 7 319
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Singh, T.J., Bhat, S.V. Morphology and conductivity studies of a new solid polymer electrolyte: (PEG)xLiClO4 . Bull Mater Sci 26, 707–714 (2003). https://doi.org/10.1007/BF02706768
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02706768