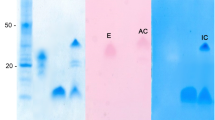
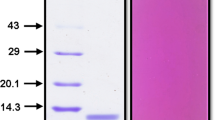
Abstract
Bowman-Birk inhibitors (BBI) isolated from plant seeds are small proteins active against trypsin and/or chymotrypsin. These inhibitors have been extensively studied in terms of their structure, interactions, function and evolution. Examination of the known three-dimensional structures of BBIs revealed similarities and subtle differences. The hydrophobic core, deduced from surface accessibility and hydrophobicity plots, corresponding to the two tandem structural domains of the double headed BBI are related by an almost exact two-fold, in contrast to the reactive site loops which depart appreciably from the two-fold symmetry. Also, the orientations of inhibitory loops in soybean and peanut inhibitors were different with respect to the rigid core. Based on the structure of Adzuki bean BBI-trypsin complex, models of trypsin and chymotryspin bound to the monomeric soybean BBI (SBI) were constructed. There were minor short contacts between the two enzymes bound to the inhibitor suggesting near independence of binding. Binding studies revealed that the inhibition of one enzyme in the presence of the other is associated with a minor negative cooperativity. In order to assess the functional significance of the reported oligomeric forms of BBI, binding of proteases to the crystallographic and non-crystallographic dimers as found in the crystal structure of peanut inhibitor were examined. It was found that all the active sites in these oligomers cannot simultaneously participate in inhibition.
Similar content being viewed by others
References
Bergeron D and Nielsen S S 1993 Partial characterization of trypsin inhibitors and N-terminal sequences of five trypsin inhibitors of great northern beans(Phaseolous vulgaris);J. Agric, Food Chem. 41 1544–1549
Chen P, Rose J, Love R, Wei A. H and Wang B C 1992 Reactive sites of an anticarcenogenic Bowman-Birk Protease inhibitor are similar to other trypsin inhibitions;J. Biol. Chem. 267 1990–1994
Cohen G H, Silverton E W and Davies D R 1981 Refined crystal structure of γ-chymotrypsin at 1.9 A resolution: Comparison with other pancreatic serine proteases;J. Mol Biol. 148 449–479
Haynes R and Feeney R E 1967 Fractionation and properties of trypsin and chymotrypsin inhibitors from lima bean;J. Biol. Chem. 242 5378–5385
Gennis L S and Cantor A. R 1976 Double-headed inhibitors from Black-eyed peas I: Purification of two new protease inhibitors and the endogenous protease by afinity chromotography;J. Biol. Chem. 251 747–753
Hummel B C W 1959 The modified spectroscopic determination of chymotrypsin, trypsin and thermolysin;Can. J. Biochem. Physiol. 37 1393–1399
Kakade M L, Simons N and Liener I E 1969 The evaluation of natural vs synthetic substrates for measuring the antitrypsic activities of soybean samples;Cereal Chem. 46 518–526
Kyte J and Doolittle R F 1982 A simple method for displaying the hydropathic character of a protein;J. Mol. Biol. 151 105–132
Laskowski M Jr and Kato I 1980 Protein inhibitors of proteinases;Annu. Rev. Biochem. 49 593–626
Lee B and Richards F M 1971 The interpretation of protein structures; estimation of static accessibilities;J. Mol. Biol. 55 379–400
Lin G, Bode W, Huber R, Chi C and Engh R A 1993.25 nm X-ray structure of the Bowman-Birk type inhibitor from mung bean in tertiary complex with porcine trypsin;Eur. J. Biochem. 212 549–555
Miller S, Janin J, Lesk A M, Chotia C 1987 Surface of monomelic proteins;J. Mol. Biol. 196 641–656
Norioka S and Ikenaka T 1983 Amino acid sequences of trypsin chymotrypsin inhibitors (A-I, A-II, B-I and B-II) from peanut (Arachis hypogaea): A discussion on molecular evolution of legume Bowman-Birk type inhibitors;J. Biochem. 94 589–599
Prakash B and Murthy M RN 1997 Source and target enzyme signature in serine protease inhibitor active site sequences;J.Biosci. 22 555–565
Prakash B, Selvaraj S, Murthy M R N, Sreerama Y N, Rama Sarma P R, Rajagopal Rao D and Gowda L R 1996 Analysis of the amino acid sequences of plant Bowman-Birk inhibitors;J. Mol. Evol. 42 560–569
Rossmann M G and Argos P 1975 A comparison of the heme binding pocket in globins and cytochrome b5;J. Biol.Chem. 250 7525–7532
Sreerama Y N, Das J R, Rao D R and Gowda L R 1997 Double headed trypsin/chymotrypsin inhibitor from horse gram—Purification, molecular and kinetic properties;J. Food. Biochem. (in press).
Suzuki A, Yamane T, Ashida T, Norioka S, Hara S and Ikenaka T 1993 Crystallographic refinement of Bowman-Birk type protease inhibitor A-II from Peanut(Arachis hypogaea) at 2-3 A resolution;J. Mol Biol. 234 722–734
Tsunogae Y, Tanaka I, Yamane T, Kikkawa J, Ashida T, Ishikawa C, Watanabe K, Nakamura S and Takahashi K 1986 Structure of trypsin binding domain of Bowman-Brik type protease inhibitor and its interaction with trypsin;J. Biochem. 100 1637–1646
Whitley E J and Bowman D E 1975 Isolation and properties of navy bean proteinase inhibitor componentI;Arch. Biochem. Biophys,169 42–50
Wu C and Whitaker Jr 1991 Homology among trypsin/chymotrypsin inhibitors from red kidney bean, Brazilian pink bean, lima bean and soybean;J. Agric. Food Chem. 39 1583–1589
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Prakash, B., Murthy, M.R.N., Sreerama, Y.N. et al. Studies on simultaneous inhibition of trypsin and chymotrypsin by horsegram Bowman-Birk inhibitor. J Biosci 22, 545–554 (1997). https://doi.org/10.1007/BF02703392
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02703392