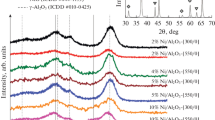
Abstract
Oxidation kinetics of niobium (columbium) in oxygen are investigated in the temperature range of 873 to 1083 K. The observed abnormal dependence of the linear oxidation rate on temperature is suggested to be due to the relative amount of NbO2 in the oxide scale. The amount of oxygen dissolved in the metal is calculated and it constitutes a small fraction of the total oxygen uptake. From hardness measurements, the oxygen diffusion coefficient in Nb can be expressed as:D = 2.72 × 10−3 exp ( − 95.4/R T) with the activation energy in kjoule/mole.
Similar content being viewed by others
References
P. Kofstad:High Temperature Oxidation of Metals, John Wiley and Sons, Inc., New York, 1966, pp. 209–218.
T. Hurlen:J. Inst. Met., 1960, vol. 89, p. 273.
D. W. Bridges and W. M. Fassell:J. Electrochem. Soc, 1956, vol. 103, p. 326.
P. Kofstad and H. Kjöllesdal:Trans. TMS-AIME, 1961, vol. 221, p. 285.
T. L. Kolski:Trans. ASM, 1962, vol. 55, p. 119.
B. B. Argent and B. Phelps:J. Inst. Met., 1960, vol. 88, p. 301.
D. W. Aylmore, S. J. Gregg, and W. B. Jepson:J. Electrochem. Soc, 1960, vol. 107, p. 495.
J. S. Sheasby, W. W. Smeltzer, and A. E. Jenkins:J. Electrochem. Soc, 1968, vol. 115, p. 338.
J. S. Sheasby:J. Electrochem. Soc, 1968, vol. 115, p. 695.
E. A. Gulbransen and D. F. Andrew:Trans. AIME, 1950, vol. 188, p. 586.
J. S. Sheasby, G. R. Wallwork, and W. W. Smeltzer:J. Electrochem. Soc, 1966, vol. 113, p. 1255.
V. K. Sikka and J. C. Rosa:Z. Metallkd., 1978, vol. 69, p. 777.
G. C. Chen and C. J. Rosa:High Temperature Science, 1980, in press.
L. Loners:Compt. Rend., 1950, vol. 231, p. 522.
P. Kofstad and S. Espevik:J. Electrochem Soc, 1965, vol. 112, p. 153.
R. A. Rapp and G. N. Goldbert:Trans. TMS-AIME, 1966, vol. 236, p. 1619.
R. Speiser and G. R. St. Pierre:The Science and Technology of Tungsten, Tantalum, Molybdenum, Niobium and Their Alloys, N. E. Promisel, ed., Pergamon Press, New York, 1964, pp. 289–330.
G. Bauer:Z. Anorg. Chem., 1941, vol. 248, p. 1.
R. P. Elliott:Constitution of Binary Alloys, First Supplement, McGraw-Hill Book Co., New York, 1965, p. 262.
N. Terao:Jpn. J. Appl. Phys., 1963, vol. 2, p. 156.
F. A. Shunk:Constitution of Binary Alloys, Second Supplement, McGraw-Hill Book Co., 1969, p. 192.
J. A. Roberson and R. A. Rapp:J. Phys. Chem. Solids, 1969, vol. 30, p. 1119.
O. Kubaschewski and B. E. Hopkins:Oxidation of Metals and Alloys, Butterworth and Co. Ltd., London, 1962, p. 9.
A. U. Seybolt:Trans. AIME, 1954, vol. 200, p. 774;J. Metals., June 1954.
R. W. Powers and M. V. Doyle:J. Appl. Phys., 1959, vol. 30, p. 514.
C. Y. Ang:Acta Metall., 1953, vol. 1, p. 123.
C. T. Sims, W. D. Klopp, and R. I. Jaffee:Trans. ASM, 1959, vol. 51, p. 256.
R. C. Weast, ed.:Handbook of Chemistry and Physics, The Chemical Rubber Co., Cleveland, OH, 1966, p. B-200.
N. B. Pilling and R. E. Bedworth:J. Inst. Met., 1923, vol. 29, p. 529.
E. A. Gulbransen and K. F. Andrew:J. Electrochem. Soc, 1958, vol. 105, p. 4.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Clenny, J.T., Rosa, C.J. Oxidation kinetics of niobium in the temperature range of 873 to 1083 K. Metall Trans A 11, 1385–1389 (1980). https://doi.org/10.1007/BF02653493
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02653493