Abstract
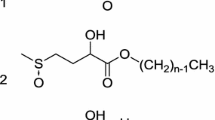
Several effective nonionic surface-active agents in which a single sucrose moiety is the only solubilizing group were described. Alkylsucroses, preparedvia sodium sucrate in dimethyl sulfoxide (DMSO) and higher alkyl bromides, are believed to be the first sugar-based surfactants which are at least equivalent to the best anionics and nonionics in cloth detergency on the basis of laboratory data; they were also active lime-soap disperstants. Another ether-linked type, the (3-alkoxy-2-hydroxypropyl) and (2-hydroxyalkyl) sucroses, derived from glycidyl ethers or olefin oxides, although second in detergency to the first type, included exceptional nonionic lathering agents, t-Dodecyl-benzylsucrose, from dodecylbenzyl chloride, was also a rather active detergent and lime-soap dispersant. Less active but easily prepared sucrose half-esters from alkenylsuccinic anhydrides were described.
One sucrose moiety in ether-linked surfactants was an effective hydrophile for alkyl hydrophobes at least as large as hexadecyl, but tridecyl derivatives were superior in activity.
Major importance was attached to the choice of solvent, catalyst, and time/temperature factors. Evidence was presented to show that sucrose derivatives were partially degraded by the alkaline conditions required in this work and that this degradation was both detrimental to detergeney and initially beneficial to lather in specific cases.
An improved procedure involving the use of DMSO for the alkaline dehydrochlorination of higher alkoxychloropropanols to glycidyl ethers was described.
Similar content being viewed by others
References
Anderson, A.W., U.S. 2,902,478 to Dow Chemical Company (1959).
Anderson, A.W., and Melstad, J.L., U.S. 2,908,681, to Dow Chemical Company (1960).
Ames, G.R., Chem. Rev.,60, 541 (1960).
Berls, R.J., U.S. 2,903,382, to Armour and Company (1959).
Black, W.A.P., Dewar, E.T., Paterson, J.C., and Rutherford, D., J. Appl. Chem.,9, 256 (1959).
Black, W.A.P., Dewar, E.T., and Rutherford, D., J. Chem. Soc., 3073 (1959).
Caldwell, C.G., and Wurzburg, O.B., U.S. 2,661,349, to National Starch Products, Inc. (1953).
Gaertner, V.R., U.S. 2,965,652, to Monsanto Chemical Company (1960).
Gaertner, V.R., and Doerr, E.L., U.S. 2,868,731, to Monsanto Chemical Company (1959).
Gaertner, V.R., U.S. 2,973,632, to Monsanto Chemical Company (1961).
Harris, J.C., “Detergency Evaluation and Testing,” Interscience Publishers Inc., New York, 1954.
Hass, H.B., Mfg. Chemist,29, 152 (1958).
Huber, W.F., and Tucker, N.B., U.S. 2.812,324, to Procter and Gamble Company (1957).
Komori, S., Okahara, M., and Okamoto, K., J. Am. Oil Chem. Soc.,37, 468 (1960).
Kononenko, O.K., and Herstein, K.M., Chem. and Eng. Data Series,1, 87 (1956).
LeMaistre, J.W., and Seymour, R.B., J. Org. Chem.,13, 782 (1948).
Lorand, E. J., U.S. 1,959,590, to Hercules Powder Company (1934).
Osipow, L.I., Snell, F.D., Marra, D., York, W.C., Finchler, A., Ind. Eng. Chem.,48, 1456 (1956); Hass, H.B., Snell, F.D., York, W.C., and Osipow, L.I., U.S. 2,893,990, to Sugar Research Foundation Inc. (1959).
Pilpel, N., Research (London),12, 68 (1959).
Reid, E.E., Ruhoff, J.R., and Burnett, R.E., in Blatt, A.H., “Organic Syntheses,” Coll. Vol. 11, John Wiley and Sons, Inc., New York, 1943, pp. 246–248.
Rhodes, C.A., Chem. Prod.,21, 320 (1958).
Winstein, S., and Henderson, R.B., in Elderfield, R.C., “Heterocyclic Compounds,” Vol. 1, John Wiley and Sons Inc., New York, 1950, p. 8.
Chem. Week,88, 45 (1961).
Author information
Authors and Affiliations
About this article
Cite this article
Gaertner, V.R. Sucrose ether- and ester-linked surfactants. J Am Oil Chem Soc 38, 410–418 (1961). https://doi.org/10.1007/BF02637976
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02637976