Abstract
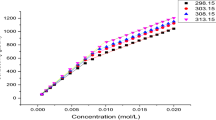
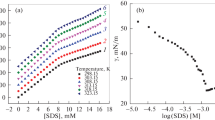
Conductivity measurements of solutions of sodium decyl sulfate and sodium dodecyl sulfate were made in water, N,N-dimethylformamide, and dimethyl sulf-oxide at 25 C. Within the concentration range 1 to 100 millimoles, the surfactants were found to form micellar aggregates. The critical micelle concentration values were found to depend on the dielectric constant of the medium and also on the hydrogen bonding nature of the solvent. The role of hydrogen bonding nature of the solvent in micelle formation is discussed.
Similar content being viewed by others

References
Becher, P., and N.K. Clifton, J. Colloid Sci. 14:519 (1959).
Sirianni, A.F., J.M.G. Cowie, and I.E. Puddington, Can. J. Chem. 40:257 (1962).
Brown, C.W., D. Cooper, and J.C.S. Moore, J. Colloid Interface Sci. 32:584 (1970).
Gopal, R., and J.R. Singh, J. Indian Chem. Soc. 49:667 (1972).
Gopal, R., and J.R. Singh, J. Phys. Chem. 77:554 (1973).
Miura, M., and M. Kodama, Bull. Chem. Soc. Jap. 45:428 (1972).
Herman, K.W., J. Phys. and Colloid Chem. 53:1 (1948).
Debye, P., Ibid. 53:1 (1948).
Nemethy, G., H.A. Scheraga, J. Chem. Phys. 36:3401 (1962).
“Hand Book of Chemistry and Physics,” 47th Edition, Chemical Rubber Co., Cleveland, OH, 1966–1967.
Dawson, L.R., in “Chemistry in Non-Aqueous Ionising Solvents,” Vol. IV, Edited by R.C. Weast, Interscience Publishers, New York, NY, 1963.
Parker, A.J., Chem. Rev. 69:1 (1969).
Kodama, M., and M. Miura, Bull. Chem. Soc. Jap. 45:2265 (1972).
Author information
Authors and Affiliations
About this article
Cite this article
Singh, H.N., Singh, S. & Tewari, K.C. Surface active anions in polar solvents: I. Conductometric studies on solutions of sodium decyl and dodecyl sulphate in water, N,N-Dimethylformamide, and dimethyl sulfoxide. J Am Oil Chem Soc 52, 436–438 (1975). https://doi.org/10.1007/BF02637483
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02637483