Abstract
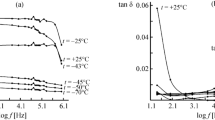
Dielectric properties (dielectric constant ε′ and dielectric loss ε″) were compared to conventional methods of analysis (viscosity, refractive index, iodine value, peroxide value and free fatty acids) for evaluating the frying quality of a blend of cottonseed and sunflower oils. The apparent relaxation time, τ, the activation energy H and the entropy change S for dielectric relaxation of the heated oil samples were calculated. Results indicate that dielectric constant and dielectric loss are useful tools for predicting deterioration occurring during heating of the oil.
Similar content being viewed by others
References
Smith, L.M., A.J. Clifford, C.L. Hamblin and R.K. Creveling,J. Am. Oil Chem. Soc. 63: 1017 (1986).
Wu, P.-F., and W.W. Nawar, Ibid.:1363 (1986).
Kupranycz, D.B., M.A. Amer, and B.E. Baker, Ibid.:332 (1986).
Porim Technology “The Practice of Frying” Palm Oil Research Institute of Malaysia by K.G. Bargen, May 1984.
Fritsch, C.W.,J. Am. Oil Chem. Soc. 58:272 (1981).
Mancini-Filho, J., L.M. Smith, R.K. Creveling and H.F. Al-Shaikh, Ibid.:1452 (1986).
El-Shami, S.M., I. Zaki Selim, I.M. El-Anwar and M.H. El-Mallah,Fat Sci. Techn., in press (1992).
Badran, B.M., I.M. El-Anwar, M.S. Ibrahim and W.M. Khalifa,J. Oil Col. Chem. Assoc. 62:199 (1979).
Petroleum Products and Lubricants, American Society for Testing Materials, Philadelphia, 1986, Method ASTM D445.
Walker, R.O. (ed.),Official and Tentative Methods of The American Oil Chemists’ Society, 3rd edn., Champaign, 1980.
Clark, F.M.,Ind. Eng. Chem. 31:327 (1939).
Balsbaugh, J.C., A.H. Howeli and A.G. Assaf,Ind. Eng. Chem. 32:1497 (1940).
El-Anwar, I.M., and S.M. Saad,Bull. NRC, Egypt 8:151 (1983).
Janicek, G., and J. Pokorny,Inst. of Chem. Tech. Pragu 44:157 (1960).
El-Anwar, I.M., and O.M. El-Nabawy,Scientific Engineering Bulletin, Part 2, 1988.
Author information
Authors and Affiliations
About this article
Cite this article
El-Shami, S.M., Selim, I.Z., El-Anwar, I.M. et al. Dielectric properties for monitoring the quality of heated oils. J Am Oil Chem Soc 69, 872–875 (1992). https://doi.org/10.1007/BF02636335
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02636335