Summary
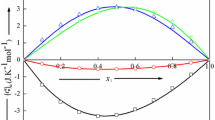
Complete binary freezing-point data have been obtained for each of the polymorphic modifications of acetamide with alpha-eleostearic acid and beta-eleostearic acid and for alpha-eleostearic acid with beta-eleostearic acid. In the light of previously published data the fact that all of these systems are of the simple eutectic type suggests that the tendency of the long-chain fatty acids to form 1∶1 molecular compounds with acetamide and with each other decreases as the degree of unsaturation increases.
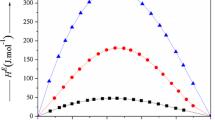
The fact that the heat of fusion of beta-eleostearic acid, 13.2 kcal. per mole, calculated from the freezing-point data is greater than that for the alpha acid, 10.5 kcal. per mole, corroborates the conclusion that the beta acid is the trans-trans-trans modification. Calculations of heats of fusion have been made, based upon all the available freezing-point data in the literature for binary systems between pairs of C18 fatty acids of different degrees of unsaturation, and certain trends have been dsicussed.
Similar content being viewed by others
References
Bickford, W. G., DuPré, E. F., Mack, C. H., and O'Connor, R. T. (in press).
de Visser, L. E. O., Rec trav. chim.,17, 182–189 (1898).
Francis, F., Collins, F. J. E., and Piper, S. H., Proc. Roy. Soc. London A,158, 691–718 (1937).
Griffiths, H. N., and Hilditch, T. P., J. Chem. Soc., 1932, 2315–2324.
Guttman, L., and Pitzer, K. S., J. Am. Chem. Soc.,67, 324–327 (1945).
Magne, F. C., and Skau, E. L., J. Am. Chem. Soc.,74, 2628–2630 (1952).
Markley, K. S., “Fatty Acids,” Interscience Publishers Inc., New York (1947), pp. 115–127.
Mod, R. R., and Skau, E. L., J. Phys. Chem.,56, 1016–1017 (1952).
Paschke, R. F., Tolberg, W., and Wheeler, D. H., J. Am. Oil Chem. Soc.,30, 97–99 (1953).
Privett, O. S., Lundberg, W. O., Khan, N. A., Tolberg, W. E., and Wheeler, D. H., J. Am. Oil Chem. Soc.,30, 61–66 (1953).
Schuette, H. A., and Vogel, H. A., Oil and Soap,17, 155–157 (1940).
Scott, R. B., Ferguson, W. J., and Brickwedde, F. G., J. Research Natl. Bur. Standards,33, 1–20 (1944).
Skau, E. L., and Saxton, B., J. Am. Chem. Soc.,50, 2693–2701 (1928).
Smith, J. C., J. Chem. Soc., 1939, 974–980.
Stewart, H. W., and Wheeler, D. H., Oil and Soap,18, 69–71 (1941).
Tipson, R. S., in Weissberer, “Technique of Organic Chemistry”, Interscience Publishers Inc., New York, 1950, vol. III, p. 425.
Todd, S. S., Oliver, G. D., and Huffman, H. M., J. Am. Chem. Soc.,69, 1519–1524 (1947).
Todd, S. S., and Parks, G. S., J. Am. Chem. Soc.,58, 134–137 (1936).
Author information
Authors and Affiliations
Additional information
One of the laboratories of the Bureau of Agricultural and Industrial Chemistry, Agricultural Research Administration, U.S. Department of Agriculture.
About this article
Cite this article
Mod, R.R., Skau, E.L. & Planck, R.W. Binary freezing-point diagrams for alpha- and beta-eleostearic acids with each other and with acetamide. J Am Oil Chem Soc 30, 368–371 (1953). https://doi.org/10.1007/BF02633772
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02633772