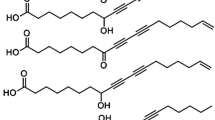
Abstract
Two heat-bodied linseed oils, with Gardner viscosities of 37 and 55 min, were saponified, converted to their methyl esters, and separated into 2 fractions with urea and methanol. Gas-liquid chromatography showed the adduct fraction, which comprised 38–41% of the total methyl esters, to contain: palmitic, stearic, oleic, “linoleic,” and trace amounts of “linolenic” acid. The nonadducting fraction (59–62%) of the total methyl esters was separated by molecular distillation at 140C/7 μ into a distillate and residue. The distillate amounted to 18–25% of the total methyl esters and had an iodine value (I.V.) of 142–145; its absorption at 232 mμ indicates 2.5–3.0% conjugated diene. Hydrogenation of this distillate gave a liquid product with an iodine number of 4 and a pour point of −50C. Gas chromatograms of the distillate and its hydrogenated derivative indicated at least 5–7 components. Comparison of these peaks with known fatty acid methyl esters indicates that the components of these fractions were either cyclic or branched esters. The nonadducting residue fraction was composed mainly of polymeric acids.
Similar content being viewed by others
References
Friedrich, J. P., Anal. Chem.33, 974, 975 (1961).
Friedrich, J. P., H. M. Teeter, J. C. Cowan, and G. E. McManis, JAOCS38, 329–332 (1961).
Greaves, J. H., and B. Laker, Chem. & Ind. (London)1961, No. 42, 1709.
Lorette, N. B., and J. H. Brown, Jr., J. Org. Chem.24, 261–262 (1959).
McDonald, J. A., JAOCS33, 394–396 (1956).
Mehta, T. N., and S. A. Sharma,Ibid. 33, 38–44 (1956).
Paschke, R. F., J. E. Jackson, and D. H. Wheeler, Ind. Eng. Chem.44, 1113–1118 (1952).
Paschke, R. F., and D. H. Wheeler, JAOCS32, 469–473 (1955).
Scholfield, C. R., and J. C. Cowan,Ibid., 36, 631–635 (1959).
Author information
Authors and Affiliations
Additional information
A laboratory of the No. Utiliz. Res. & Dev. Div., ARS, U.S.D.A.
About this article
Cite this article
Gast, L.E., Schneider, W.J., Forest, C.A. et al. Composition of methyl esters from heat-bodied linseed oils. J Am Oil Chem Soc 40, 287–289 (1963). https://doi.org/10.1007/BF02633694
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02633694