Summary
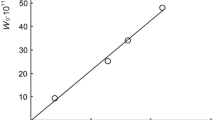
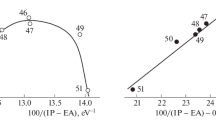
The kinetics of a Diels-Alder-type reaction between diethylazodicarboxylate andt,t-9,11-octadecadienoic acid has been studied in various solvent systems and with acidic catalysts. The rate of the reaction was found to vary as follows: a) the reaction rate is faster in polar solvents than in nonpolar solvents; b) addition of acidic catalysts to nonpolar solvents increases the rate of the reaction, and acidic catalysts appear to have no effect on the rate of reaction in polar solvents; c) when compatibility can be maintained, addition of water to polar solvent systems appears to increase the reaction rate in proportion to the amount of water added.
The reaction follows second-order or pseudo second-order kinetics. Probably it is more complex than the over-all reaction kinetics indicate. Certain reactions were studied at two temperatures, and information on activation energy of the reaction has been obtained.
Similar content being viewed by others
References
Diels, O., Blum, J. H., and Koll, W., Ann.,443, 242 (1925).
Getman, E. H., and Daniels, F., “Physical Chemistry,” 7th ed., p. 361, London, John Wiley and Sons, 1945.
Kass, J. P., and Burr, G. O., J. Am. Chem. Soc.,61, 3292 (1939).
Mangold, C., Monatsh,15, 309 (1894).
Nichols, P. L., Herb, S. F., and Riemenschneider, R. W., J. Am. Chem. Soc.,73, 247 (1951).
Rodgman, A., Wright, G. F., J. Org. Chem.,18, 480 (1953).
Author information
Authors and Affiliations
About this article
Cite this article
Gast, L.E., Bell, E.W. & Teeter, H.M. Reactions of conjugated fatty acids. III. Kinetics of the Diels-Alder reaction. J Am Oil Chem Soc 33, 278–281 (1956). https://doi.org/10.1007/BF02630861
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02630861