Summary
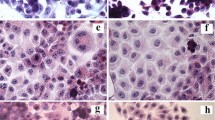
A rat liver-derived epithelial cell line transformed withdl-ethionine and the corresponding control cell line were characterized according to morphological and cytochemical criteria to establish their origin from liver epithelium and to identify cellular changes due to transformation bydl-ethionine. The presence of intermediate junctions confirms the epithelial nature; glycogen accumulation and glucose-6-phosphatase activity confirm the hepatic origin of the cells. Persistent alterations resulting from ethionine transformation were variations in cell shape and size, focal multilayered growth, an increase in the nucleolar: nuclear ratio, and a reduction in the number of cells displaying a primary cilium. Hyperplasia of the inner nuclear membrane, elongation and branching of mitochondria, and a reduction in the length and frequency of cell junctions were also characteristic of the transformed cells.
Similar content being viewed by others
References
Farber, E. Ethionine carcinogenesis. Adv. Cancer Res. 7: 383–474; 1963.
Smith, R. C.; Salmon, W. D. Formation of S-adenosylethionine by ethionine-treated rats. Arch. Biochem. Biophys. 111: 191–196; 1965.
Farber, E.; Shull, K. H.; Villa-Trevino, S.; Lombardi, B.; Thomas, M. Biochemical pathology of acute hepatic adenosinetriphosphate deficiency. Nature 203: 34–40; 1964.
Shull, K. H.; McConomy, J.; Vogt, M.; Castillo, A.; Farber, E. On the mechanism of induction of hepatic adenosine triphosphate deficiency by ethionine. J. Biol. Chem. 241: 5060–5070; 1966.
Okazaki, K.; Shull, K. H.; Farber, E. Effects of ethionine on adenosine triphosphate levels and ionic composition of liver cell nuclei. J. Biol. Chem. 243: 4661–4666; 1968.
Cox, R.; Irving, C. C. Inhibition of DNA methylation by S-adenosylethionine with the production of methyl-deficient DNA in regenerating rat liver. Cancer Res. 37: 222–225; 1977.
Christman, J. K.; Price, P.; Pedrinam, L.; Acs, G. Correlation between hypomethylation of DNA and expression of globin genes in Friend erythroleukemia cells. Eur. J. Biochem. 81: 53–61; 1977.
Boehm, T. L. J.; Drahovsky, D. Effect of carcinogen ethionine on enzymatic methylation of DNA sequences with various degrees of repetitiveness. Eur. J. Cancer 15: 1167–1173; 1979.
Wildenauer, D.; Gross, H. J. Methyldeficient mammalian 4S RNA: evidence for L-ethionine-induced inhibition ofN 6-dimethyladenosine synthesis in rat liver tRNA. Nucleic Acids Res. 12: 279–288; 1974.
Friedman, S. Alterations of tRNA modification in mammalian systems: the effect of ethionine. Nucleic Acids Res. 4: 1853–1871; 1977.
Swann, P. F.; Peacock, A. C.; Bunting, S. Carcinogenesis and cellular injury, the effect of ethionine on ribonucleic acid synthesis in rat liver. Biochem. J. 150: 334–344; 1975.
Wolf, S. F.; Schlessinger, D. Nuclear metabolism of ribosomal RNA in growing, methionine limited, and ethionine-treated HeLa cells. Biochemistry 16: 2783–2791; 1977.
Goswami, B. B.; Sharma, O. K. Accumulation of methyl-deficient rat liver messenger ribonucleic acid on ethionine administration. Biochemistry 19: 2101–2198; 1980.
Tuck, M. T.; Cox, R. Ethionine inhibitsin vivo methylation of nuclear proteins. Carcinogenesis 3: 431–434; 1982.
Stekol, J. A.; Weiss, S.; Somerville, C. A study of the comparative metabolism of ethionine and methionine in the male and female rat. Arch. Biochem. Biophys. 100: 86–90; 1963.
Rosen, L. Ethylationin vivo of purines in rat-liver tRNA byl-ethionine. Biochem. Biophys. Res. Commun. 33: 546–550; 1968.
Ortwerth, B. J.; Novelli, G. D. Studies on the incorporation ofl-ethionine-ethyl-l-14C into the transfer RNA of rat liver. Cancer Res. 29: 380–390; 1969.
Pegg, A. E. Studies of the ethylation of rat liver transfer ribonucleic acid after administration ofl-ethionine. Biochem. J. 128: 59–68; 1972.
Friedman, M.; Shull, K. H.; Farber, E. Highly selectivein vivo ethylation of rat liver nuclear protein. Biochem. Biophys. Res. Commun. 34: 857–864; 1969.
Wilson, M. J.; Hatfield, D. L.; Poirier, L. A. Aminoacylation of ethionine to rat liver tRNAMet and its incorporation into protein. FEBS Lett. 128: 157–160; 1981.
Brown, J. D.; Wilson, M. J.; Poirier, L. A. Neoplastic conversion of rat liver epithelial cells in culture by ethionine and S-adenosylethionine. Carcinogenesis 4: 173–177; 1983.
Idoine, J. B.; Elliott, J. M.; Wilson, M. J.; Weisburger, E. K. Rat liver cells in culture: effects of storage, long-term culture, and transformation on some enzyme levels. In Vitro 12: 541–553; 1976.
Moorhead, P. S.; Nowell, P. C. Chromosome cytology. Methods Med. Res. 10: 310–322; 1964.
Coolidge, B. J.; Howard, R. M. Animal histology procedures of the pathological technology section of the National Cancer Institute. Bethesda, MD: U. S. Department of Health, Education and Welfare; 1979: 59–67; 142.
Lillie, R. D. Histopathologic techic and practical histochemistry. New York: McGraw-Hill Book Company; 1965: 312–324.
Rutenberg, A. M.; Fischbein, J. W.; Hanker, J. S.; Wasserkrug, H. L.; Seligman, A. M. Histochemical and ultrastructural demonstration of γ-glutamyl-transpeptidase activity. J. Histochem. Cytochem. 17: 517–526; 1969.
Albrecht-Buehler, G.; Bushnell, A. The ultrastructure of primary cilia in quiescent 3T3 cells. Exp. Cell Res. 126: 427–437; 1980.
Ginzburg, I.; Cornelis, P.; Giveon, D.; Littauer, U. Functionally impaired tRNA from ethionine-treated rats as detected in injectedXenopus oocytes. Nucleic Acids Res. 6: 657–672; 1979.
Borenfreund, E.; Higgins, P. J.; Steinglass, M.; Bendick, A. Properties and malignant transformation of established rat liver parenchymal cells in culture. J. Natl. Cancer Inst. 55: 375–384; 1975.
Schaeffer, W. I.; Heintz, N. H. A diploid rat liver cell culture. IV. Malignant transformation by aflatoxin B1. In Vitro 14: 418–427; 1978.
Herring, A. S.; Raychandhur, R.; Kelley, S. P.; Iype, P. T. Repeated establishment of diploid epithelial cell cultures from normal and partially hepatomized rats. In Vitro 19: 576–588; 1983.
Yoshimura, H.; Harris, R.; Yokoyama, S.; Takahashi, S.; Sells, M. A.; Pan, S. F.; Lombardi, B. Anaplastic carcinomas in nude mice and in original donor strain rats inoculated with cultured oval cells. Am. J. Pathol. 110: 322–331; 1983.
Tucker, R. W.; Pardee, A. B. Centriole ciliation is related to quiescence and DNA synthesis in 3T3 cells. Cell 17: 527–535; 1979.
Parry, E. W. Membrane-bounded intracellular structures in hepatocytes after exposure to sodium tetraphenyl boron. J. Pathol. 104: 210–212; 1971.
Kuhn, C. Structure of bronchio-alveolar cell carcinoma. Cancer 30: 1107–1118; 1972.
Franke, W. W.; Scherr, U. Structures and functions of the nuclear envelope. Busch, H. ed. The cell nucleus, Vol. 1. New York: Academic Press; 1974: 219–347.
Puvion, E.; Viron, A.; Bernhard, W. Unusual accumulation of ribonucleoprotein constituents in the nucleus of cultured rat liver cells after hypothermal shock. Biol. Cell. 29: 81–88; 1977.
Heine, U.; Sverak, L.; Kondratick, J.; Bonar, R. A. The behavior of HeLa-S3 cells under the influence of supranormal temperatures. J. Ultrastruct. Res. 34: 375–396; 1971.
Shinozuka, H. P.; Goldblatt, P. J.; Farber, E. The disorganization of hepatic cell nucleoli induced by ethionine and its reversal by adenine. J. Cell Biol. 36: 313–328; 1968.
Puvion, E.; Puvion-Dutilleul, F.; Leduc, E. H. The formation of nucleolar perichromatin granules. J. Ultrastruct. Res. 76: 181–190; 1981.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Heine, U.I., Wilson, M.J. & Munoz, E.F. Characterization of rat liver cells transformed in culture bydl-ethionine. In Vitro 20, 291–301 (1984). https://doi.org/10.1007/BF02618591
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02618591