Abstract
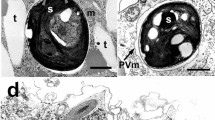
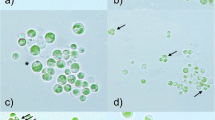
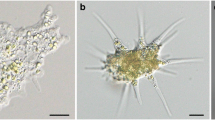
Gram-negative bacteria 4.5–5.5 μm in length and 1.2 μm in diameter are found in gastrodermal cells of three stains of freshwater green hydras,Hydra viridis (Ohio and Carolina from North America, Jubilee strain from England). They are motile via single polar flagella. They were detected in live animals, Jensen stained material, and electron micrographic sections. Bacteria lose motility quickly upon release from hydra cells. Green hydras harbor strain-specific numbers of chlorellae in these cells. Other tissue types lack algae. The chlorella-hydra symbiosis can be disassociated and the partners grown separately; transfer of photosynthate from algae to hydra occurs. Here we report the presence of endocellular bacterial vesicles specifically associated with cells that contain the symbiotic chlorellae. No cells that contained algae and lacked bacteria were seen. Vesicles, especially conspicuous in sexually mature green hydras, are probably produced upon extrusion from the cell. They contain either algae and bacteria or bacteria alone and are often expelled to the surrounding medium via the coelenteron. Bacteria are absent in nerve cells, interstitial cells, nematocysts, mucous cells, sperm, and probably in most of the other cell types that lack algae. They are present in at least one cell type that lacked algae: the columnar ovarian cells. Bacteria were lost in “bleached” hydras, those induced to lose their algae by high intensity light in a solution of DCMU, a standard inhibitor of photosynthesis. They were absent in a fourth strain of green hydra (Connecticut Valley,H. viridis) and inH. fusca andH. littoralis, two freshwater nonsymbiotic hydras. All of the hydra lacking bacteria contain conspicuous lipid droplets in their cells. The presence of large numbers of bacteria has implications for interpretations of metabolic exchange between host and algal symbionts and for extrapolation of metabolic data from strain to strain ofH. viridis.
Similar content being viewed by others
Literature Cited
Cernichiari, E., Muscatine, L., Smith, D. C. 1969. Maltose excretion by the symbiotic algae ofHydra viridis. Proceedings of the Royal Society of London B Biological Sciences173:557–576.
Cook, C. 1972. Benefit to symbiotic zoochlorella from feeding by green hydra. Biological Bulletin142:236–242.
Cooper, G., Margulis, L. 1978. Delay in migration of symbiotic algae inHydra viridis by inhibitors of microtubule protein polymerization. Cytobios19:7–19.
David, C. 1973. A quantitative method for maceration of hydra tissue. Wilhelm Roux’ Archiv fuer Entwicklungsmechanik der Organismen171:259–268.
Geddes, P. 1882. The yellow cells of radiolarians and coelenterates. Proceedings of the Royal Society of Edinburgh11:377–396.
Muscatine, L. 1965. Symbiosis of hydra and algae III. Extracellular products of the algae. Comparative Biochemistry and Physiology16:77–92.
Muscatine, L., Lenhoff, H. 1965. Symbiosis of hydra and algae I. Effects of some environmental cations on growth of symbiotic and aposymbiotic hydra. Biological Bulletin129:316–328.
Nicholls K. 1978. Jensen staining, a neglected tool in Phycology. Transactions of the American Microscopical Society97:129–132.
Oschman, J. 1967. Structure and reproduction of the algal symbionts ofHydra viridis. Journal of Phycology3:221–228.
Pardy, R. 1976. The production of aposymbiotic green hydra by the photodestruction of their symbiotic algae. Biological Bulletin151:225–235.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Margulis, L., Thorington, G., Berger, B. et al. Endosymbiotic bacteria associated with the intracellular green algae ofHydra viridis . Current Microbiology 1, 227–232 (1978). https://doi.org/10.1007/BF02602848
Issue Date:
DOI: https://doi.org/10.1007/BF02602848