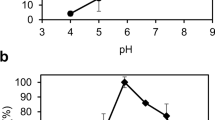
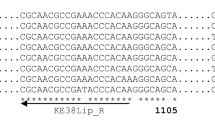
Abstract
ARhizomucor miehie cDNA library constructed inEscherichia coli was screened with synthetic oligonucleotides designed from knowledge of a partial amino acid sequence of the secreted triglyceride lipase (triacyl-glycerol acylhydrolase EC 3.1.1.3) from this fungus. Lipase-specific recombinants were isolated and their insert sequenced. Unlike characterized bacterial and mammalian triglyceride lipases, the fungal enzyme is synthesized as a precursor, including a 70 amino acid residue propeptide between the 24 amino acid residues of the signal peptide and the 269 residues of the mature enzyme. The precursor processing mechanism, which involves cleavage between a methionine and a serine residue, is unknown. By sequence comparison with other lipases, a serine residue involved in substrate binding was identified in the fungal lipase. The sequence around this residue is well-conserved among characterized lipases. Conservation of an intron in an isolated cDNA recombinant and immunoprecipitation of in vitro synthesizedR. miehei translation products indicates that the expression of the lipase gene might involve inefficient mRNA splicing.
Similar content being viewed by others
Abbreviations
- RML:
-
Rhizomucor miehei lipase
- SDS:
-
sodium dodecylsulphate
- bp:
-
base pair
- HIC:
-
hydrophobic interaction chromatography
- HPLC:
-
high performance liquid chromatography
- kb:
-
kilobase
- LU:
-
lipase units.
References
Schipper, M.A.A. (1978)Studies in Mycology, No. 17, pp. 53–71, Institute of the Royal Netherlands Academy of Sciences and Letters, Amsterdam, The Netherlands.
Cooney, D.G., and Emerson, R. (1964)Thermophilic fungi, pp. 17–27, W.H. Freeman and Company, San Francisco, CA.
Moskowitz, G.J., Cassaigne, R., West, I.R., Shen, T., and Feldman, L.I. (1977)J. Agric. Food Chem. 25, 1146–1150.
Huge-Jensen, B., Galluzzo, D.R., and Jensen, R.G. (1987)Lipids 22, 559–565.
Iwai, M., and Tsujiska, Y. (1974)Agric. Biol. Chem. 38, 1241–1247.
Iwai, M., Tsujiska, Y., and Tominaga, Y. (1979)Agric. Biol. Chem. 43, 893–900.
Benzonana, G. (1974)Lipids 9, 166–172.
Brockman, H.L. (1981)Methods Enzymol. 71, 619–627.
Lowry, O.H., Rosebrough, N.J., Farr, A.L., and Randall, R.J. (1951)J. Biol. Chem. 193, 265–275.
Dubois, M., Gilles, K.A., Hamilton, J.K., Rebers, P.A., and Smith, F. (1956)Anal. Chem. 28, 350–356.
Rüegg, U.Th., and Rudinger, J. (1974)Israel. J. Chem. 12, 391–401.
Friedman, M., Krull, L.H., and Cavins, J.E. (1970)J. Biol. Chem. 245, 3868–3871.
Moody, A.J., Thim, L., and Valverde, I. (1984)FEBS Lett. 172, 142–148.
Thim, L., Hansen, M.T., and Sørensen, A.R. (1987)FEBS Lett. 212, 307–312.
Boel, E., Beck, A.-M., Randrup, K., Dræger, B., Fiil, N.P., and Foltmann, B. (1986)Proteins 1, 363–369.
Boel, E., Hjort, I., Svensson, B., Norris, F., Norris, K.E., and Fiil, N.P. (1984)Eur. Mol. Biol. Org. J. 3, 1097–1102.
Boel, E., Vuust, J., Norris, F., Norris, K., Wind, A., Rehfeld, J.F., and Marcker, K.A. (1983)Proc. Natl. Acad. Sci. USA 80, 2866–2869.
Gergen, J.P., Stern, R.H., and Wensink, C. (1979)Nucl. Acids Res. 7, 2115–2135.
Maxam, A., and Gilbert, W. (1980)Methods Enzymol. 65, 499–560.
von Heijne, G. (1986)Nucl. Acids Res. 14, 4683–4690.
Kugimiya, W., Otani, Y., Hashimoto, Y., and Takagi, Y. (1986)Biochem. Biophys. Res. Commun. 141, 185–190.
Lee, C.Y., and Iandolo, J.J. (1984)J. Bacteriol. 166, 385–391.
Götz, F., Popp, F., Korn, E., and Schleifer, K.H. (1985)Nucl. Acids Res. 13, 5895–5906.
Docherty, A.J.P., Bodmer, M.W., Angal, S., Verger, R., Riviero, C., Lowe, P.A., Lyons, A., Emtage, J.S., and Harris, T.J.R. (1985)Nucl. Acids Res. 13, 1891–1903.
De Caro, J., Boudouard, M., Bonicel, J., Guidoni, A., Desnuelle, P., and Rovery, M. (1981)Biochim. Biophys. Acta 671, 129–138.
McLean, J., Fielding, C., Drayna, D., Dieplinger, H., Baer, B., Kohr, W., Henzel, W., and Lawn, R. (1986)Proc. Natl. Acad. Sci. USA 83, 2335–2339.
Wion, K.L., Kirchgessner, T.G., Lusis, A.J., Schotz, M.C., and Lawn, R.M. (1987)Science 235, 1638–1641.
Guidoni, A., Benkouka, F., De Caro, J., and Rovery, M. (1981)Biochim. Biophys. Acta 660, 148–150.
Sternby, B., Engström, Aa., Hellman, U., Vikert, A.M., Sternby, N.-H., and Borgström, B. (1984)Biochim. Biophys. Acta 784, 75–80.
Schwarz, T. (1986)FEBS Lett. 200, 1–10.
Boel, E., Hansen, M.T., Hjort, I., Høgh, I., and Fiil, N.P. (1984)Eur. Mol. Biol. Org. J. 3, 1581–1585.
Ballance, D.J. (1986)Yeast 2, 229–236.
Author information
Authors and Affiliations
About this article
Cite this article
Boel, E., Huge-Jensen, B., Christensen, M. et al. Rhizomucor miehei triglyceride lipase is synthesized as a precursor. Lipids 23, 701–706 (1988). https://doi.org/10.1007/BF02535672
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02535672