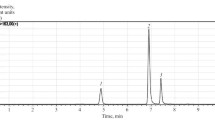
Abstract
Plant sterols and triterpenes exhibit a wide range of pharmacological activities. As part of our ongoing studies of the medicinal aspects of Maritime flora, particularly the herbal remedies of the Micmac and Malecite Indians, we determined the nature of the sterols and triterpenes ofTanacetum vulgare L. (Compositae)—a widely used herbal remedy usually referred to as tansy. By using thin layer and gas chromatographics, nuclear magnetic resonance (NMR) spectroscopy and combined gas chromatography-mass spectrometry, we were able to identify β-sitosterol as the major sterol and α-amyrin as the major triterpene of tansy. We also identified the sterols stigmasterol, campesterol and cholesterol, and the triterpenes β-amyrin and taraxasterol. A fourth triterpene was tentatively identified as pseudo-taraxasterol. The successful therapeutic application of this herb may be due partly to the presence of one or more of these compounds. The sterols and triterpenes of tansy have not been previously reported; neither, to our knowledge, have the NMR spectra of the amyrins and the NMR and mass spectra of taraxasterol.
Similar content being viewed by others
References
Chandler, R.F., Freeman, L., and Hooper, S.N. (1979) J. Ethnopharm. 1, 49–68.
Chandler, R.F., and Hooper, S.N., Can. J. Pharm. Sci., in press.
Favreau, G., and Mockle, J.A. (1962) Can. Pharm. J. 95, 32–34.
Kasprzyk, Z., and Kozierowska, T. (1966) Bull. Acad. Pol. Sci., Ser. Sci. Biol. 14, 645–649.
Matthes, H., and Serger, H. (1909) Arch. Pharm. 247, 418–431.
Vogel, V.J. (1970) in American Indian Medicine, p. 380, University of Oklahoma Press, Norman, OK.
Moerman, D.E. (1977) American Medical Ethnobotany, pp. 188–189, Garland Publishing, New York, NY.
Hutchens, A.R., Tretchikoff, N.G., and Tretchikoff, N.K. (1973) Indian Herbalogy of North America, pp. 279–280, Merco, Windsor, Canada.
Millspaugh, C.F. (1974) American Medicinal Plants, pp. 338–343, Dover Publications, New York, NY.
Grieve, M. (1974) A Modern Herbal, pp. 789–790, Hafner Press, New York, NY.
Levy, J. de B. (1966) Herbal Handbook for Everyone, p. 125, Faber and Faber, London.
Izvestiya Akademii Nauk Armyanskoi SSR. Biologischeskie Nauki. Armenia, Russia 7 27 (1954); as cited in Willaman, J.J., and Li, H.L. (1970) Lloydia 33(3A), 81.
Rudloff, E. Van, and Underhill, E.W. (1965) Phytochemistry 4, 11–17.
Banthorpe, D.V., and Mann, J. (1972) Phytochemistry 11, 2589–2591.
Nano, G.M., Bicchi, C., Frattini, C., and Gallino, M. (1979) Planta Med. 35, 270–274.
Tetenyi, P., Kaposi, P., and Hethelyi, E. (1975) Phytochemistry 14, 1539–1544.
Samek, Z., Holub, M., Grabarczyk, H., Drozdz, B., and Herout, V. (1973) Collec. Czech. Chem. Commun. 38, 1971–1976.
Yunusov, A.I., Sidyakin, G.P., and Nigmatullaev, A.M. (1979) Chem. Nat. Compd. USSR 15, 88–89.
Wagner, H., Flores, G., and Geissman, T.A. (1972) Phytochemistry 11, 451.
Ban'kovskii, A.I., Glyzin, V.I., and Smirnova, L.P. (1978) Farmatsiya 27, 24–28 (Chem. Abstr. 89, 72955a).
Banthorpe, D.V., and Wirz-Justice A. (1972) J. Chem. Soc., Perkin Trans. 1, 1769–1772.
Schulte, K.E., Ruecker, G., and Dressen, F.P. (1966) Planta Med. 14, 162–170.
Hartwell, J.L., and Abbott, B.J. (1969) Adv. Pharmacol. Chemother. 7, 117–209.
Hartwell, J.L. (1976) Cancer Treatment Rep. 60, 1031–1067.
Miles, D.H., Kokpol, U., Zalkow, L.H., Steindel, S.J., and Nabors, J.B. (1974) J. Pharm. Sci. 63, 613–615.
Pettit, G.R. (1977) Biosyn. Prod. Cancer Chemother. 1, 61–96, 177–186.
Pettit, G.R., and Cragg, G.M. (1978) Biosyn. Prod. Cancer Chemother. 2, 11–60, 121–126.
Pettit, G.R., and Ode, R.H. (1979) Biosyn. Prod. Cancer Chemother. 3, 9–18, 65–124.
Chandler, R.F., Hooper, S.N., and Ismail, H.A. (1979) Can. J. Pharm. Sci. 14, 61–62.
Subbiah, M.T.R. (1973) Am. J. Clin. Nutr. 26, 219–225.
Chaturvedi, A.K., Parmar, S.S., Nigam, S.K., Bhatnagar, S.C., Misra, G., and Sastry, B.V.R. (1976) Pharmacol. Res. Commun. 8, 199–210.
Farnsworth, N.R., and Cordell, G.A. (1976) Lloydia 39, 420–424.
Benoit, P.S., Fong, H.H.S., Svoboda, G.H., and Farnsworth, N.R. (1976) Lloydia 39, 160–171.
Shibata, S. (1977) in New Natural Products and Plant Drugs with Pharmacological, Biological or Therapeutic Activity (Wagner, H., and Wolff, P., eds.) pp. 177–196, Springer-Verlag, New York, NY.
Krochmal, A., and Krochmal, C. (1973) A Guide to the Medicinal Plants of the United States, pp. 212–213, Quadrangle, New York, NY.
Lewis, W.H., and Elvin-Lewis, M.P.F. (1977) Medical Botany Plants Affecting Man's Health, pp. 58, 84, 370, Wiley, New York, NY.
The Pharmacopeia of the United States of America, 6th revision, (1882) pp. 316–317, William Wood and Co., New York, NY.
Kingsbury, J.M. (1964) Poisonous Plants of the United States and Canada, p. 437, Prentice-Hall, Englewood Cliffs, NJ.
Hardin, J.W., and Arena, J.M. (1974) Human Poisoning from Native and Cultivated Plants, 2nd edn., pp. 151–152, Duke University Press, Durham, NC.
Claus, E.P. (1956) Pharmacognosy, 3rd edn., p. 316, Lea and Febiger, Philadelphia, PA.
Albert-Puleo, M. (1978) Econ. Bot. 32, 65–74.
Hussey, J.S. (1974) Econ. Bot. 28, 311–337.
Mechling, W.H. (1959) Anthropologica 8, 239–263.
Method Ca 6b-53 (1972) The Official and Tentative Methods of the American Oil Chemists' Society, 3rd edn., American Oil Chemists' Society Champaign, IL.
Safe, L.M., Wong, C.J., and Chandler, R.F. (1974) J. Pharm. Sci. 63, 464–466.
Madrigal, R.V., Plattner, R.D., and Smith, C.R., Jr. (1975) Lipids 10, 208–213.
Vroman, H.E., and Cohen, C.F. (1967) J. Lipid Res. 8, 150–152.
Morris, L.J. (1966) J. Lipid Res. 7, 717–732.
Fong, H.H.S. (1973) in Experiments in the Pharmaceutical Biological Sciences (Malone, M.H., and McLaughlin, J.L., eds.) pp. 43–51, American Association of Colleges of Pharmacy, Stockton, CA.
Hooper, S.N., and Chandler, R.F. (1978) J. Pharm. Sci. 67, 1157–1159.
Shamma, M., Glick, R.E., and Mumma, R.O. (1962) J. Org. Chem. 27, 4512–4517.
Bhacca, N.S., and Williams, D.H. (1964) Applications of NMR Spectroscopy in Organic Chemistry, pp. 13–41, 77–89, Holden-Day, San Francisco, CA.
Budzikiewicz, H., Wilson, J.M., and Djerassi, C. (1963) J. Am. Chem. Soc. 85, 3688–3699.
Budzikiewicz, H., Wilson, J.M., and Djerassi, C. (1964) Structure Elucidation of Natural Products by Mass Spectrometry, Vol. II. Steroids, Terpenoids, Sugars and Miscellaneous Classes, pp. 121–140, Holden-Day, San Francisco, CA.
Author information
Authors and Affiliations
About this article
Cite this article
Chandler, R.F., Hooper, S.N., Hooper, D.L. et al. Herbal remedies of the maritime Indians: Sterols and triterpenes ofTanacetum vulgare L. (Tansy). Lipids 17, 102–106 (1982). https://doi.org/10.1007/BF02535183
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02535183