Abstract
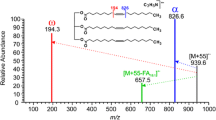
Chemical ionization (CI) mass spectra of long-chain saturated, monenoic and dienoic wax ester analogues, using isobutane and methane as reagent gases, are reported and compared. Because less fragmentation occurs, CI spectra are simpler than the electron impact spectra. The quasi molecular ion (M+1) is the base peak in the spectra. Saturated, monenoic and dienoic wax ester mixtures have been analyzed by this technique.
Similar content being viewed by others
References
Ryhage, R., and Stenhagen, E. (1960) J. Lipid Res. 1, 361–390.
Aasen, A.J., Hofstetter, H.H., Ijengar, B.I., and Holman, R.T. (1971) Lipids 6, 502–507.
Spencer, G.F., Plattner, R.D., and Miwa, T.K. (1977) J. Am. Oil Chem. Soc. 54, 187–189.
Spencer, G.F. (1979) J. Am. Oil Chem. Soc. 56, 642–646.
Vajdi, M., Nawar, W.W., and Merritt, C. (1981) J. Am. Oil Chem. Soc. 58, 106–110.
Author information
Authors and Affiliations
Additional information
The mention of firm names or trade products does not imply that they are endorsed or recommended by the U.S. Department of Agriculture over other firms or similar products not mentioned.
About this article
Cite this article
Plattner, R.D., Spencer, G.F. Chemical lonization mass spectrometry of wax esters. Lipids 18, 68–73 (1983). https://doi.org/10.1007/BF02534693
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02534693