Abstract
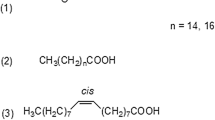
The proton magnetic resonance spectra of isomeric triglycerides of isovaleric and palmitic acids in the presence of the downfield and upfield chemical shift reagents Eu(fod)3 and Pr(fod)3 were studied. The resonance profiles of the γ-protons on the isovaleroyl chains are distinctive for each of the four possible triglyceride isomers at low shift reagent/triglyceride ratios. With either reagent, the well defined γ-methyl isovalerate doublets can be used to identify 2-isovaleroyl and 1,3-isovaleroyl structures and to analyze isomeric mixtures. This technique will be useful for positional analysis of natural cetacean triglycerides containing isovaleric acid.
Similar content being viewed by others
References
Hopkins, C.Y., Prog. Chem. Fats Other Lipids 8:215 (1966).
Swern, D., and J.P. Wineburg, JAOCS 48:371 (1971).
Wineburg, J.P., and D. Swern, Ibid. 49:267 (1972).
Pfeffer, P.E., and H.L. Rothbart, Tetrahedron Lett. 25:2533 (1972).
Almqvist, S.O., R. Andersson, Y. Shahab, and K. Olsson, Acta Chem. Scand. 26:3378 (1972).
Pfeffer, P.E., and H.L. Rothbart, Ibid. 27:3131 (1973).
Wedmid, Y., C. Litchfield, R.G. Ackman, J.C. Sipos, C.A. Eaton, and E.D. Mitchell, Biochim. Biophys. Acta 326:439 (1973).
Varanasi, U., and D.C. Malins, Ibid. 231:415 (1971).
Blomberg, J., Lipids 9:461 (1974).
Selinger, Z., and Y. Lapidot, J. Lipid Res. 7:174 (1966).
Kester, E.B., C.J. Gaiser, and M.E. Lazar, J. Org. Chem. 8:550 (1943).
Privett, O.S., and L.J. Nutter, Lipids 2:149 (1967).
Mitchell, L.C., JAOCS 49:281 (1972).
Mattson, F.H., R.A. Volpenhein, and J.B. Martin, J. Lipid Res. 5:374 (1964).
Author information
Authors and Affiliations
About this article
Cite this article
Wedmid, Y., Litchfield, C. Positional analysis of isovaleroyl triglycerides using proton magnetic resonance with Eu(fod)3 and Pr(fod)3 shift reagents: I. Model compounds. Lipids 10, 145–151 (1975). https://doi.org/10.1007/BF02534152
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02534152