Abstract
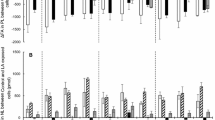
The metabolic fate ofcis-positional isomers of octadecenoates has been compared to that of naturally occurring oleic acid (cis-Δ9). Radioactive mixtures of tritium-labeled positional octadecenoate isomer and oleic acid-10-14C were administered to laying hens, and their eggs were analyzed for the isotopic ratios (3H/14C) incorporated into total egg lipid, triglycerides, and phospholipids. Variations in the isotopic ratios indicated the comparative metabolic utilization ofcis-positional isomers Δ8 through Δ12. Incorporation into egg lipid fractions is as follows: triglycerides: Δ9>Δ8, Δ9>Δ10, Δ9>Δ11, Δ9>12; phospholipid: Δ9>Δ8, Δ9>Δ10, Δ9<Δ11, Δ9<Δ12.
Similar content being viewed by others
References
Scholfield, C.R., V.L. Davison, and H.J. Dutton, JAOCS 44:648 (1967).
Carpenter, D.L., and H.T. Slover, Ibid. 50:372 (1973).
Reitz, R.C., M. ElSheikh, W.E.M., Lands, I.A. Ismail, and F.D. Gunstone, Biochim. Biophys. Acta 176:480 (1969).
Lippel, K., D.L. Carpenter, F.D. Gunstone, and I.A. Ismail, Lipids 8:124 (1973).
Sgoutas, D.S., Biochemistry 9:1826 (1970).
Goller, H.J., D.S. Sgoutas, I.A. Ismail, and F.D. Gunstone, Ibid. 9:3072 (1970).
Mounts, T.L., E.A. Emken, W.K. Rohwedder, and H.J. Dutton, Lipids 6:912 (1971).
Emken, E.A., C.R. Scholfield, and H.J. Dutton, JAOCS 41:388 (1964).
Chang, Huei-Chi, J. Janke, F. Pusch, and R.T. Holman, Biochim. Biophys. Acta 306:21 (1973).
Mounts, T.L., and H.J. Dutton, Biochim. Biophys. Acta 431:9 (1976).
Mattson, F.H., and R.A. Volpenhein, J. Lipid Res. 2:58 (1961).
Robertson, A.F., and W.E.M. Lands, Biochemistry 1:804 (1962).
Christie, W.W., and J.H. Moore, Biochim. Biophys. Acta 218:83 (1970).
Author information
Authors and Affiliations
About this article
Cite this article
Mounts, T.L. Double bond position affects metabolism ofcis-octadecenoates. Lipids 11, 676–679 (1976). https://doi.org/10.1007/BF02532885
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02532885