Summary
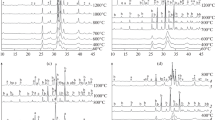
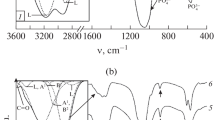
A method is described for determination of the titratable hydroxide ion in calcium phosphate precipitates. The procedure requires accurate analysis of the other titratable species in the crystal lattice but is unaffected by the presence of other lattice constituents or impurities. The method was applied to precipitates that had been previously analyzed by solution thermodynamic techniques, and the results were consistent with the earlier observations. The hydroxide content of the precipitates increased with crystal maturity and with increasing pH of the precipitation medium. The hydroxide content of the amorphous phase and the immediate post-amorphous-crystalline transformation phase was shown to be nearly zero. After 3 to 4 days' maturation, the hydroxide content of precipitates prepared at pH values of 7, 8, and 9 was shown to increase to approximately 23, 40, 56% of that required for pure hydroxyapatite.
Similar content being viewed by others
References
Arnold, P.W.: The nature of precipitated calcium phosphates, Trans. Faraday Soc.40:1061–1072, 1950
Brown, W.E., Lehr, J.R., Smith, J.P., and Frazier, A.W.: Crystallography of octacalcium phosphate, J. Am. Chem. Soc.79:5318–5319, 1957
Posner, A.S., Fabry, C., Dallemagne, M.J.: Defect apatite series in synthetic and natural calcium phosphates: the concept of pseudoapatites, Biochim. Biophys. Acta15:304–305, 1954
Posner, A.S., Perloff, A.: Apatites deficient in divalent cations, J. Res. Natl. Bur. Stand.58:279–286, 1957
Winand, L., Dallemagne, M.J., Duyckaerts, G.: Hydrogen bonding in apatitic calcium phosphates, Nature190:164–165, 1961
Kuhl, G., Nebergall, W.H.: Hydrogenphosphat- und carbonatapatite, Z. Anorg. Allgem. Chem.324:313–320, 1963
Berry, E.E.: The structure and composition of some calcium-deficient apatites, J. Inorgan. Nucl. Chem.29:317–327, 1967
Berry, E.E.: The structure and composition of some calcium-deficient apatites—II, J. Inorgan. Nucl. Chem.29:1585–1590, 1967
Berry, E.E.: The structure and composition of some calcium-deficient apatites, Bull. Soc. Chim. Fr. (Spec. No.) 1765–1770, 1968
Bett, J.A.S., Christner, L.G., Hall, W.K.: Studies of the hydrogen held by solids. XII. Hydroxyapatite catalysts, J. Am. Chem. Soc.89:5535–5541, 1967
Cant, N.W., Bett, J.A.S., Wilson, G.R., Hall, W.K.: The vibrational spectrum of hydroxyl groups in hydroxyapatites, Spectrochimica Acta27A:425–439, 1971
Joris, S.J., Amberg, C.H.: The nature of deficiency in non-stoichiometric hydroxyapatites. II. Spectroscopic studies of calcium and strontium hydroxyapatites, J. Phys. Chem.75:3172–3178, 1971
Termine, J.D., Lundy, D.R.: Hydroxide and carbonate in rat bone mineral and its synthetic analogues, Calcif. Tissue Res.13:73–82, 1973
Blumenthal, N.D., Posner, A.S.: Hydroxyapatite: mechanism of formation and properties, Calcif. Tissue Res.13:235–243, 1973
Vatassery, G.T., Armstrong, W.D., Singer, L.: Determination of hydroxyl content of calcified tissue mineral, Calcif. Tissue Res.5:183–188, 1970
Vatassery, G.T., Armstrong, W.D., Singer, L.: The hydroxyl content of calcified tissue mineral; comment to the letter of Blitz and Pellegrino, Calcif. Tissue Res.7:264–266, 1971
Biltz, R.M., Pellegrino, E.D.: The hydroxyl content of calcified tissue mineral, Calcif. Tissue Res.7:259–263, 1971
Meyer, J.L., Eanes, E.D.: A thermodynamic analysis of the secondary transition in the spontaneous precipitation of calcium phosphate, Calcif. Tissue Res.25:209–216, 1978
Fowler, B.O.: Infrared studies of apatites. II. Preparation of normal and isotopically substituted calcium, strontium, and barium hydroxyapatites and spectra-structure-composition correlations, Inorgan. Chem.13:207–214, 1974
Murphy, J., Riley, J.P.: A modified single solution method for the determination of phosphate in natural waters, Anal. Chim. Acta27:31–36, 1962
Hirschman, A., Sobel, A.E.: Composition of the mineral deposited during in vitro calcification in relation to the fluid phase, Arch. Biochem. Biophys.110:237–243, 1965
Gee, A., Deitz, V.R.: Pyrophosphate formation upon ignition of precipitated basic calcium phosphates, J. Am. Chem. Soc.77:2961–2965, 1955
Bates, R.G.: First dissociation constant of phosphoric acid from 0° to 60° C; limitations of the electromotive force method for moderately strong acids, J. Res. Natl. Bur. Stand.47:127–134, 1951
Bates, R.G., Acree, S.F.: ΔH values of certain phosphate-chloride mixtures, and the second dissociation constant of phosphoric acid from 0° to 60° C, J. Res. Natl. Bur. Stand.30:129–155, 1943
Harned, H.S., Davis, R.: The ionization constant of carbonic acid in water and the solubility of carbon dioxide in water and aqueous salt solutions from 0 to 50°, J. Am. Chem. Soc.65:2030–2037, 1943
Chughtai, A., Marshall, R., Nancollas, G.H.: Complexes in calcium phosphate solutions, J. Phys. Chem.72:208–211, 1968
Davies, C.W.: Ion Association. Butterworth and Co., London, 1962
Bevington, P.R.: Data Reduction and Error Analysis for the Physical Sciences, p. 56. McGraw-Hill, New York, 1969
Eanes, E.D., Meyer, J.L.: The maturation of crystalline calcium phosphates in aqueous suspensions at physiologic pH, Calcif. Tissue Res.23:259–269, 1977
Meyer, J.L., Eanes, E.D.: A thermodynamic analysis of the amorphous to crystalline calcium phosphate transformation, Calcif. Tissue Res.25:59–68, 1978
Greenfield, D.J., Eanes, E.D.: Formation chemistry of amorphous calcium phosphates prepared from carbonate containing solutions, Calcif. Tissue Res.9:152–162, 1972
Elliott, J.C.: The problems of the composition and structure of the mineral components of the hard tissues, Clin. Orthop.93:313–345, 1973
Termine, J.D., Eanes, E.D.: Comparative chemistry of amorphous and apatitic calcium phosphate preparations, Calcif. Tissue Res.10:171–179, 1972
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Meyer, J.L. Hydroxyl content of solution-precipitated calcium phosphates. Calcif Tissue Int 27, 153–160 (1979). https://doi.org/10.1007/BF02441178
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02441178