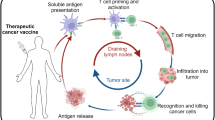
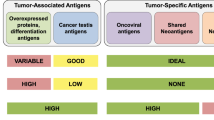
Abstract
Background: Tumor-specific cytotoxic T-lymphocytes (CTLs) can be isolated from the solid tumors, draining lymph nodes, metastatic effusions, and peripheral blood of cancer patients. Despite this evidence for a cell-mediated immune response to cancer, attempts at active specific immunotherapy using cancer vaccines have met with little success in clinical trials.
Methods: We have reviewed the immunobiology of the cell-mediated immune response to cancer by focusing on what is known about the major histocompatibility complex (MHC)-restricted interaction between tumor cells and CD8+ or CD4+ T-cells. In addition, we review the recent advances in the identification of tumor-associated antigens (TAAs) that are recognized by tumor-specific CTLs in melanoma and other cancers. In discussing these antigens, we highlight the recent identification of several MHC-restricted antigenic peptides that are recognized by CTLs from patients with melanoma and those with ovarian and breast cancer. We examine the implications that the discovery of these TAAs and peptides will have on the development of new anticancer vaccines. We review the most recent vaccine trials in melanoma and other cancers and focus on current concepts aimed at improving the therapeutic efficacy of future vaccines, including genetically engineered tumor cell vaccines.
Conclusions: With the recent identification of several TAAs and antigenic peptide epitopes in melanoma and other cancers, immunotherapy researchers are now focusing on new strategies for the development of anticancer vaccines. As the repertoire of known TAAs increases and our understanding of the immunobiology of cell-mediated immunity to cancer improves, immunotherapists remain cautiously optimistic in their quest for effective cancer vaccines.
Article PDF
Similar content being viewed by others
References
Gross L. Intradermal immunization of C3H mice against a sarcoma that originated in an animal of the same line.Cancer Res 1943;3:326–33.
Prehn RT, Main JM. Immunity to methylcholanthrene-induced sarcomas. J Natl Cancer Inst 1957;18:769–78.
Oettgen HF, Old LJ. In: DeVita VI, Hellman S, Rosenberg SA, eds.Biologic therapy of cancer. Philadelphia: JB Lippincott, 1991.
Ostrand-Rosenberg, S. Tumor immunotherapy: the tumor cell as an antigen-presenting cell.Curr Opin Immunol 1994;6:722–7.
Greenberg PD, Cheever MA, Fefer A. Therapy of disseminated murine leukemia with cyclophosphamide and immune Lyt-1+, 2− T cells.J Exp Med 1985;161:1122–34.
Wang P, Vanky F, Klein E. MHC class-I-restricted auto-tumor-specific CD4+ CD8− T-cell clones established from autologous mixed lymphocyte-tumor-cell culture.Int J Cancer 1992;51:962–7.
Topalian SL, Rivoltini L, Mancini M, Ng J, Hartzman RJ, Rosenberg SA. Melanoma-specific CD4+ T-lymphocytes recognize human melanoma antigens processed and presented by Epstein-Barr virus-transformed B-cells.Int J Cancer 1994;58:69–79.
Topalian SL, Rivoltini L, Mancini M, et al. Human CD4+ T-cells specifically recognize a shared melanoma-associated antigen encoded by the tyrosinase gene.Proc Natl Acad Sci U S A 1994;91:9461–5.
Goedegebuure PS, Harel W, LeMay LG, Kan-Mitchell J, Mitchell MS. Cytotoxic CD4+ lymphocyte clones reactive with melanoma: the role of HLA and accessory molecules.Vaccine Res 1993;2:227–34.
Chen Q, Hersey P. MHC-restricted responses of CD8+ and CD4+ T-cell clones from regional lymph nodes of melanoma patients.Int J Cancer 1992;51:218–24.
Hom SS, Topalian SL, Simonis T, Mancini M, Rosenberg SA. Common expression of melanoma tumor-associated antigens recognized by human tumor-infiltrating lymphocytes: analysis by human leukocyte antigen restriction.J Immunol 1991;10:153–64.
Peoples GE, Schoof DD, Andrews VR, Goedegebuure PS, Eberlein TJ. T-cell recognition of ovarian cancer.Surgery 1993;114:227–34.
Schendel DJ, Gansbacher B, Oberneder R, et al. Tumor-specific lysis of human renal cell carcinoma by tumor infiltrating lymphocytes: HLA-A2-restricted recognition of autologous and allogeneic tumor lines.J Immunol 1993;151:4209–20.
van der Bruggen P, Traversari C, Chomez P, et al. A gene encoding an antigen recognized by cytolytic T-lymphocytes on a human melanoma.Science 1991;254:1643–7.
van der Bruggen P, Szikora J-P, Boel P, et al. Autologous cytolytic T-lymphocytes recognize a MAGE-1 nonapeptide on melanomas expressing HLA-Cw1601.Eur J Immunol 1994;24:2134–40.
Gaugler B, Van den Eynde B, van der Bruggen P, et al. Human gene MAGE-3 codes for an antigen recognized on a melanoma by autologous cytotoxic T-lymphocytes.J Exp Med 1994;179:921–30.
Wolfel T, Van Pel A, Brichard V, et al. Two tyrosinase nonapeptides recognized on HLA-A2 melanomas.Eur J Immunol 1994;24:759–64.
Cox AL, Skipper J, Chen Y, et al. Identification of a peptide recognized by five melanoma-specific human cytotoxic T-cell lines.Science 1994;264:716–9.
Henderson RA, Cox AL, Sakaguchi K, Appella E, Shabanowitz J, Hunt DF, Engelhard VH. Direct identification of an endogenous peptide recognized by multiple HLA-A2.1-specific cytotoxic T-cells.Proc Natl Acad Sci U S A 1993;90:10275–80.
Kawakami Y, Eliyahu S, Sakaguchi K, et al. Identification of the immunodominant peptides of the MART-1 human melanoma antigen recognized by the majority of HLA-A2-restricted tumor infiltrating lymphocytes.J Exp Med 1994;180:347–52.
Peoples GE, Goedegebuure PS, Smith R, Linehan DC, Yoshino I, Eberlein TJ. Breast and ovarian cancer-specific cytotoxic T-lymphocytes recognize the same HER2/neu derived peptide.Proc Natl Acad Sci U S A 1995;92:432–6.
Kotera Y, Fontenot JD, Pecher G, Metzgar RS, Finn OJ. Humoral immunity against a tandem repeat epitope of human mucin MUC-1 in sera from breast, pancreatic and colon cancer patients.Cancer Res 1994;54:2856–60.
Kawakami Y, Eliyahu S, Delgado CH, et al. Cloning of the gene coding for a shared human melanoma antigen recognized by autologous T-cells infiltrating into tumor.Proc Natl Acad Sci U S A 1994;91:3515–9.
Yoshino I, Peoples GE, Goedegebuure PS, Schoof DD, Eberlein TJ. The sensitivity of human ovarian epithelial tumor cells to cytotoxic T-cells is associated with the expression of HER2/neu.J Immunol 1994;152:2393–400.
Peace DJ, Smith JW, Chen W, You SG, Cosand WL, Blake J, Cheever MA. Lysis of ras oncogene-transformed cells by specific cytotoxic T-lymphocytes elicited by primary in vitro immunization with mutated ras peptide.J Exp Med 1994;179:473–9.
Berzofsky JA. Epitope selection and design of synthetic vaccines: molecular approaches to enhancing immunogenicity and cross-reactivity of engineered vaccine.Ann N Y Acad Sci 1993;12:256–64.
Brichard V, Van Pel A, Wolfel T, et al. The tyrosinase gene codes for an antigen recognized by autologous cytolytic T-lymphocytes on HLA-A2 melanomas.J Exp Med 1993;178:489–95.
Ada GL. In: Paul WE, ed.Fundamental immunology. New York: Raven, 1993:1329.
Morton DL, Foshag LJ, Hoon DS, et al. Prolongation of survival in metastatic melanoma after active specific immunotherapy with a new polyvalent melanoma vaccine.Ann Surg 1992;216:463–82.
Mitchell MS, Harel W, Kempf RA, et al. Active-specific immunotherapy for melanoma.J Clin Oncol 1990;8:856–69.
Berd D, Maguire HC Jr, McCue P, Mastrangelo MJ. Treatment of metastatic melanoma with an autologous tumor-cell vaccine: clinical and immunologic results in 64 patients.J Clin Oncol 1990;8:1858–67.
Sivanandham M, Wallack MK. Clinical trials with VMO for melanoma.Ann N Y Acad Sci 1993;690:178–89.
Bystryn JC. Immunogenicity and clinical activity of a polyvalent melanoma antigen vaccine prepared from shed antigens.Ann N Y Acad Sci 1993;690:190–203.
Livingston PO, Wong GY, Adluri S, et al. Improved survival in stage III melanoma patients with GM2 antibodies: a randomized trial of adjuvant vaccination with GM2 ganglioside.J Clin Oncol 1994;12:1036–44.
Allison AC, Byars NE. Immunologic adjuvants; desirable properties and side effects.Mol Immunol 1991;28:279–84.
Pardoll DM, Tumor antigens: a new look for the 1990's.Nature 1994;369:357–8.
Rosenberg SA, Packard BS, Aebersold PM, Solomon D, et al. Use of tumor infiltrating lymphocytes and interleukin-2 in the immunotherapy of patients with metastatic melanoma.N Engl J Med 1988;319:1676–80.
Legha SS. Interferons in the treatment of malignant melanoma. A review of recent trials.Cancer 1986;57:1675–7.
Fearon ER, Pardoll DM, Itaya T, et al. Interleukin-2 production by tumor cells bypass T helper function in the generation of an antitumor response.Cell 1990;60:397–403.
Gansbacher B, Zeier K, Daniels B, Cronin K, Bennerji R, Gilboa E. Interleukin 2 gene transfer into tumor cells abrogates tumorigenicity and induces protective immunity.J Exp Med 1990;172:1217–24.
Tepper RI, Pattengall PK, Leder P. Murine interleukin-4 displays potent antitumor activity in vivo.Cell 1989;57:503–12.
Porgador A, Tzehoval E, Katz E, et al. Interleukin-6 transfection into Lewis lung carcinoma tumor cells suppresses the malignant phenotype and confers immunotherapeutic competence against parental metastatic cells.Cancer Res 1992;52:3679–86.
Tahara H, Zeh HJ, Storkus WJ, et al. Fibroblasts genetically engineered to secrete interleukin 12 can suppress tumor growth and induce antitumor immunity to a murine melanoma in vivo.Cancer Res 1994;54:182–9.
Blankenstein T, Qin Z, Uberla K, et al. Tumor suppression after tumor cell targeted tumor necrosis factor-a gene transfer.J Exp Med 1991;173:1047–53.
Dranoff G, Jaffee E, Lazenby A, et al. Vaccination with irradiated tumor cells engineered to secrete murine granulocyte-macrophage colony stimulating factor stimulates potent, specific and long-lasting anti-tumor immunity.Proc Natl Acad Sci USA 1993;90:3539–3543.
Finn OJ. Breast cancer immunotherapy. Massachusetts Department of Public Health Breast Cancer Research Lecture Series, Dana Farber Cancer Institute, Boston, MA, November 8, 1994.
Srivastava PK, Udono H. Heat-shock protein-peptide complexes in cancer immunotherapy.Curr Opin Immunol 1994;6:728–32.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Linehan, D.C., Goedegebuure, P.S. & Eberlein, T.J. Vaccine therapy for cancer. Annals of Surgical Oncology 3, 219–228 (1996). https://doi.org/10.1007/BF02305804
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02305804