Abstract
Background: The overexpression of heat shock protein 27 (hsp-27) in early-stage breast cancer is associated with histopathologic features of poor prognosis and clinically with an increased probability of disease recurrence. Hsp-27 is overexpressed in 25% of invasive ductal carcinomas (IDC); however, its distribution in ductal carcinoma in situ (DCIS) and DCIS associated with IDC has not been investigated. We postulated that hsp-27 might be detected and variably expressed in DCIS and, like HER-2/neu oncoprotein expression, might be a tumor-specific marker worthy of future clinical investigation.
Methods: To test these hypotheses, the distribution of hsp-27 in noncomedo and comedo DCIS, and DCIS associated with IDC, was evaluated by immunohistochemistry and compared with HER-2/neu expression within the same cancers.
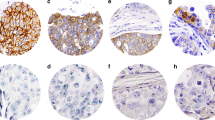
Results: Hsp-27 was overexpressed in 28 of 47 (∼60%) cases of DCIS; expression in pure DCIS was 16 of 24 (67%), and 12 of 23 (∼50%) in DCIS associated with IDC. Hsp-27 expression by in situ and invasive components of the same neoplasm were concordant in 22 of 23 (∼95%) cases tested. Comedo variants appeared to have somewhat higher hsp-27 expression than noncomedo DCIS, whether or not there was an associated IDC. These results are reminiscent of HER-2/neu oncoprotein expression in DCIS and DCIS associated with IDC observed by others. However, although 4 of 22 (18%) cancers containing DCIS + IDC expressed HER-2/neu, no relationship with hsp-27 expression in the same cancers was observed.
Conclusions: We found a high incidence of hsp-27 overexpression in DCIS and in DCIS associated with IDC. This rate is twice that previously observed in IDC alone. Hsp-27 expression is independent of HER-2/neu expression.
Similar content being viewed by others
References
Chaudhuri B, Crist KA, Mucci S, Malafa M, Chaudhuri PK. Distribution of estrogen receptor in ductal carcinoma in situ of the breast.Surgery 1993;113:134–7.
Carper SW, Rocheleau TA, Storm FK. cDNA sequence of a human heat shock protein hsp-27.Nucleic Acids Res 1990;18:6457.
Edwards DP, Adams DJ, McGuire WL. Estradiol stimulates synthesis of a major intracellular protein in the human breast cancer cell line MCF-7.Breast Cancer Res Treat 1981;1:209–24.
Carper SW, Duffy JJ, Gerner EW. Heat shock proteins in thermotolerance and other cellular processes.Cancer Res 1987;47:5249–55.
Lindquist S, Craig EA. The heat-shock proteins.Annu Rev Genet 1988;22:631–77.
Hickey E, Brandon SE, Potter R, Stein G, Stein J, Weber LA. Sequence and organization of genes encoding the human 27 kDa heat shock protein.Nucleic Acids Res 1986;14:4127–45.
Adams DJ, Hajj H, Edwards DP, Bjercke RJ, McGuire WL. Detection of a Mw 24,000 estrogen-regulated protein in human breast cancer by monoclonal antibodies.Cancer Res 1983;43:4297–301.
Chamness GC, Ruiz A, Fulcher L, Clark G, Fuqua SAW, McGuire WL. Estrogen-inducible heat shock protein 27 predicts recurrence in node-negative breast cancer [Abstract].Proc AACR 1989;30:252.
Tandon AK, Clark GM, Chamness GC, et al. Heat shock/stress response proteins: biological and clinical significance in breast cancer [Abstract].Proc ASCO 1990;9:23.
Allred DC, Clark GM, Tandon AK, et al. HER-2/neu in node-negative breast cancer: prognostic significance of overexpression influenced by the presence of in situ carcinoma.J Clin Oncol 1992;10:566–605.
Tetu B, Lacasse B, Bouchard H-L, Lagace R, Huot J, Landry J. Prognostic significance of hsp-27 expression in malignant fibrous histiocytoma: a clinicopathological and immunohistochemical study.Cancer Res 1992;52:2325–8.
Storm FK, Mahvi DM, Gilchrist KW. Hsp-27 is not expressed and has no prognostic significance in rectal adenocarcinoma.Reg Cancer Treat 1993;3:146–8.
Storm FK, Mahvi DM, Gilchrist KW. Hsp-27 has no diagnostic or prognostic significance in prostate or bladder cancer.Urology 1993;42:379–82.
Page DL, Anderson TJ.Diagnostic histopathology of the breast. Edinburgh: Churchill Livingstone, 1987.
Thor A, Benz C, More D, et al. Stress response protein (srp-27) determination in primary human breast carcinomas: clinical, histologic, and prognostic correlations.J Natl Cancer Inst 1991;83:170–8.
Allred DC, Clark GM, Molina R, et al. Overexpression of HER-2/neu and its relationship with other prognostic factors change during the progression of in situ to invasive breast cancer.Hum Pathol 1992;23:974–9.
Schmitt ST, Silen N, Sadowski NL, Connolly JL, Harris JR. Ductal carcinoma in situ (intraductal carcinoma) of the breast.N Engl J Med 1988;318:898–903.
Page DL, Dupont WD, Rogers LW, Landenberger M. Intraductal carcinoma of the breast: follow-up after biopsy only.Cancer 1982;49:751–8.
Recht A, Solin LJ, Kurtz JM, et al. Conservative surgery and radiotherapy (CS and RT) for ductal carcinoma in situ (DCIS) [Abstract].Proc Am Soc Clin Oncol 1991;10:47.
Holland R, Hendricks JHCL, Verbeek ALM, Mravunac M, Schuurman S, Stekhovan JH. Extent of distribution and mammographic histologic correlation of breast ductal carcinoma in situ.Lancet 1991;335:519–22.
Ciocca DR, Adams DJ, Edwards DP, Bjercke RJ, McGuire WL. Distribution of an estrogen-induced protein with a molecular weight of 24,000 in normal and malignant human tissues and cells.Cancer Res 1983;43:1204–10.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Storm, F.K., Gilchrist, K.W., Warner, T.F.C.S. et al. Distribution of hsp-27 and HER-2/neu in in situ and invasive ductal breast carcinomas. Annals of Surgical Oncology 2, 43–48 (1995). https://doi.org/10.1007/BF02303701
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02303701