Abstract
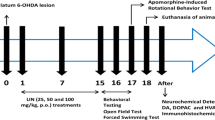
Phospholipase A2 (PLA2) is a key enzyme in the phospholipid metabolism. In the CNS intracellular PLA2 plays an essential role in signal transduction by affecting both dopamine (DA) release and DA-receptor sensitivity. In schizophrenia a disordered phospholipid metabolism and increased activity of PLA2 have been reported. In this study we investigated the effects of intracerebral PLA2 injections on dopaminergic neurotransmission in rats using Ungerstedt's model of rotational behavior. Circling behavior induced by the DA agonist apomorphine after unilateral PLA2 injections into the substantia nigra pars compacta was recorded. Seven and 21 days after intranigral PLA2 injection, apomorphine induced an ipsilateral rotation indicating a long-lasting inhibition of ipsilateral nigrostriatal dopaminergic pathway by PLA2 application. In schizophrenia a reduced dopaminergic activity in the frontal cortex has been hypothesized. Recent spectroscopy studies reported on an accelerated breakdown of membrane phospholipids in the frontal cortex from schizophrenics. The present findings suggest that increased PLA2 activity in schizophrenia could accelerate the breakdown of membrane phospholipids and thus contribute to a hypodopaminergy in the frontal cortex of schizophrenic patients.
Similar content being viewed by others
References
Anand-Srivastava MB, Johnson RA 81981) Role of phospholipids in coupling of adenosine and dopamine receptors to striatal adenylate cyclase. J Neurochem 36: 1819–1828
Bradford PG, Marinetti GV, Abood LG (1983) Stimulation of phospholipase A2 and secretion of catecholamines from brain synaptosomes by potassium and A23187. J Neurochem 41: 1684–1693
Cadet JL, Hu M, Jackson-Lewis V (1989) Behavioral and biochemical effects of intranigral injection of phospholipase-A2. Biol Psychiatry 26:106–110
Carman LS, Gage FH, Shults CW (1991) Partial lesion of the substantia nigra: relation between extent of lesion and rotational behavior. Brain Res 553:275–283
De Groot J (1959) The rat forebrain in stereotaxic coordinates. Verhandeligen der koninklijke Nederlandse Akademie van Wetenschappen, afd. Natuurkunde. N. V. Noord-Hollandische Uitgevers Maatschappij, Amsterdam
Deicken RF, Merrin E, Calabrese G, Dillon W, Weiner MW, Fein G (1993)31Phosphorous MRSI of the frontal and parietal lobes in schizophrenia. Biol Psychiatry 33:46A
Deicken RF, Merrin EL, Floyd TC, Weiner MW (1995) Correlation between left frontal phospholipids and Wisconsin Card Sort Test performance in schizophrenia. Schizophr Res 14:177–181
Di Chiara G, Porceddu ML, Morelli M, Mulas ML, Gessa GL (1979) Substnatia nigra as an out-put station for striatal dopaminergic responses: role of a GABA-mediated inhibition of pars reticulata neurons. Naunyn Schmiedebergs Arch Pharmacol 306:153–159
Farooqui AA, Hirashima Y, Horrocks LA (1992) Brain phospholipases and their role in signal transduction. In: Bazan NG, Murphy MG, Tofano G (eds) Neurobiology of essential fatty acids. Plenum Press, New York, pp 11–26
Gattaz WF, Köllisch M, Thuren T, Virtanen JA, Kinnunen PKJ (1987) Increased plasma phospholipase-A2 activity in schizophrenic patients: reduction after neuroleptic therapy. Biol Psychiatry 22:421–426
Gattaz WF, Hübner CK, Nevalainen TJ, Thuren T, Kinnunen PKJ (1990) Increased serum phospholipase A2 activity in schizophrenia: a replication study. Biol Psychiatry 28:495–501
Gattaz WF, Schmitt A, Maras A (1995) Increased platelet phospholipase A2 activity in schizophrenia. Schizophr Res 16:1–6
Noponen M, Sanfilipo M, Samanich K, Ryer H, Ko G, Angrist B, Wolkin A, Rotrosen J (1993) Elevated PLA2 activity in schizophrenics and other psychiatric patients. Biol Psychiatry 34: 641–649
Ohmichi M, Hirota K, Koike K, Kadowaki K, Miyake A, Kiyama H, Tohyama M, Tanizawa O (1989) Involvement of extracellular calcium and arachidonate in [3H] dopamine release from rat tuberoinfundibular neurons. Neuroendocrinology 50:481–487
Olianas MC, De Montis GM, Concu A, Tagliamonte A, Di Chiara G (1978) Intranigral kainic acid: evidence for nigral nondopaminergic neurons controlling posture and behavior in a manner opposite to the dopaminergic ones. Eur J Pharmacol 49: 223–232
Oliveira CR, Duarte EP, Carvalho AP (1984) Effects of phospholipase digestion and lysophosphatidylcholine on dopamine receptor binding. J Neurochem 43:455–465
Pangerl AM, Steudle A, Jaroni HW, Rüfer R, Gattaz WF (1991) Increased platelet membrane lysophosphatidylcholine in schizophrenia. Biol Psychiatry 30:837–840
Pettegrew JW, Keshavan MS, Minshew NJ (1993)31P nuclear magnetic resonance spectroscopy: neurodevelopment and schizophrenia. Schizophr Bull 19:35–53
Reid MS, Herrera-Marschitz M, Hökfelt T, Lindefors N, Persson H, Ungerstedt U (1990) Striatonigral GABA, dynorphin, substance P and neurokinin A modulation of nigrostriatal dopamine release: evidence for direct regulatory mechanisms. Exp Brain Res 82:293–303
Rotrosen J, Wolkin A (1987) Phospholipid and prostaglandin hypotheses of schizophrenia. In: Meltzer HY (ed) Psychopharmacology: the third generation of progress. Raven Press, New York, pp 759–764
Ungerstedt U, Arbuthnott GW (1970) Quantitative recording of rotational behavior in rats after 6-hydroxydopamine lesions of the nigrostriatal dopamine system. Brain Res 24:485–493
Weinberger DR (1987) Implications of normal brain development for the pathogenesis of schizophrenia. Arch Gen Psychiatry 44: 660–669
Williamson P, Drost D, Stanley J, Carr T, Morrison S, Merskey H (1991) Localized phosphorous 31 magnetic resonance spectroscopy in chronic schizophrenic patients and normal controls. Arch Gen Psychiatry 48:578
Zetterström T, Herrera-Marschitz M, Ungerstedt U (1986) Simultaneous measurement of dopamine release and rotational behaviour in 6-hydroxy-dopamine denervated rats using intracerebral dialysis. Brain Res 376:1–7
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brunner, J., Gattaz, W.F. Intracerebral injection of phospholipase A2 inhibits dopamine-mediated behavior in rats: Possible implications for schizophrenia. Eur Arch Psychiatry Clin Nuerosci 246, 13–16 (1995). https://doi.org/10.1007/BF02191810
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02191810