Abstract
Methanol oxidation on chromium-molybdenum oxide catalysts with an atom ratio Mo/Cr=0.4–6.5 proceeds via the scheme:
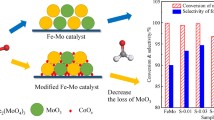
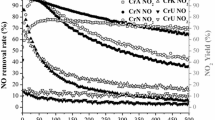
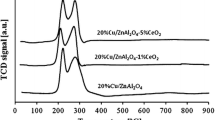
The samples with an atom ratio Mo/Cr=1.6–1.9 have the maximum activity and selectivity. Activity, selectivity and kinetic regularities of these samples and of commercial iron-molybdenum catalysts are similar.
Abstract
Окисление метанола на хромомолибденовых окисных катализаторах с атомным отношением Mo/Cr=0,4–6,5 идет по схеме:
Максималяную активность и избирательность имеют образцы с атомным отношением Mo/Cr=1,6–1,9. Активность, избирательностя и кинетические закономености этих образцов и промышленного железомолибденового катализатора близки между собой.
Similar content being viewed by others
References
N. Pernikone, J. Liberti, L. Ersini: In Osnovy predvidenia kataliticheskogo deistvia, Vol. 1, pp. 244–251. Proc. IVth International Congress on Catalysis, Izd. Nauka, Moscow 1970.
Yu.I. Yermakov: ibid.In, pp. 272–274. Proc. IVth International Congress on Catalysis, Izd. Nauka, Moscow 1970.
G.D. Kolovertnov, G.K. Boreskov, V.A. Dzis'ko, B.I. Popov, D.V. Tarasova, G.G. Belugina: Kinet. Katal.,6, 1052 (1965).
V.N. Bibin, B.I. Popov: Kinet. Katal.,9, 618 (1968);10, 1326 (1969).
G.K. Boreskov, B.I. Popov, V.N. Bibin, E.S. Kozishnikova: Kinet. Katal.,9, 796 (1968).
B.I. Popov, T.S. Popov, G.N. Kustova, L.G. Karakchiev, L.M. Plyasova: “Inorganic materials” (in press).
B.I. Popov, K.D. Osipova: Kinet. Katal.,12, 731 (1971).
B.I. Popov, K.D. Osipova, Yu.D. Pankratiev: Kinet. Katal.,12, 1243 (1971).
B.I. Popov, K.D. Osipova, V.F. Malakhov, A.M. Kolchin: Kinet. Katal.,12, 1464 (1971).
B.I. Popov. V.N. Bibin, G.K. Boreskov: Kinet. Katal. (in press).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Popov, T.S., Popov, B.I., Bibin, V.N. et al. Catalytic properties of chromium-molybdenum oxide catalysts in methanol oxidation. React Kinet Catal Lett 3, 169–175 (1975). https://doi.org/10.1007/BF02187510
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02187510