Abstract
Thermal decompositions of zinc(II) chloroacetate and its complexes with nicotinamide and caffeine were studied by means of TG/DTG, DTA, IR and mass spectroscopy. Thermal analysis showed that presence of the halogen significantly influenced the thermal decomposition. Decompositions may be characterized as two step reactions (release of nicotinamide or caffeine followed by pyrolysis of the carboxylate anion). Zinc chloride, CO, CO2, CH2O, ClCH2CHO were found in gaseous products of the thermal decomposition.
Similar content being viewed by others
References
P. Baraldi, Spectrochim. Acta, 35A (1979) 1006.
V. V. Panevčik and V. M. Gorjajev, Chimija i Chim. Technol., 22 (1979) 1303.
V. V. Panevčik, V. M. Gorjajev and V. G. Guslev, Žurnal Neorg. Chim., 27 (1982) 2199.
K. Györyová, V. Balek and V. Zeleňák, Thermochim. Acta, 234 (1994) 221.
K. Györyová and V. Balek, J. Thermal Anal., 40 (1993) 519.
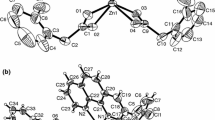
V. Zeleňák, K. Györyová, I. Císařová and J. Loub, Submitted to Acta Cryst., C.
Powder Diffraction File, 5-0664; ICPDS.
C. D. Rao, B. Paul, B. K. Mohapatra and S. Guru, J. Inorg. Nucl. Chem., 40 (1978) 134.
X.-M. Chen and T. C. W. Mak, Inorg. Chim. Acta, 182 (1991) 139.
A. Civadze, G. Cinciadze, N. Gongadze and J. Charitonov, Koord. Khim., 9 (1975) 1221.
N. N. Greenwood, E. J. F. Ross and B. P. Straughan, Index of Vibrational Spectra of Inorganic and Organometallic Compounds, Vol. 1, Butterworth London 1992.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zeleňák, V., Györyová, K. & Simon, J. Thermal properties of zinc(II) chloroacetate and its complexes with nicotinamide and caffeine. Journal of Thermal Analysis 46, 573–579 (1996). https://doi.org/10.1007/BF02135037
Issue Date:
DOI: https://doi.org/10.1007/BF02135037