Abstract
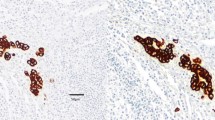
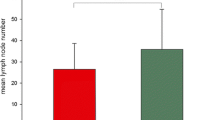
PURPOSE: Whereas lymph node metastases in colorectal carcinoma are an important prognostic factor, the prognostic relevance of occult tumor cells in lymph nodes is not elucidated at present. Therefore, our study intended to assess the rate of patients with occult tumor cells in histopathologically negative lymph nodes. Furthermore, we tried to evaluate an eventual influence of these occult tumor cells on patients' prognoses. METHODS: For examination, we used paraffin blocks of lymph nodes, tumor-negative by conventional histopathology, from 49 patients with colorectal carcinoma (Stage I–III) after a curative (RO) tumor resection in 1987. After preparation of tissue blocks using the serial sectioning technique, the specimens were stained with the alkaline phosphatase, antialkaline phosphatase method and two monoclonal antibodies (AE1/AE3 and Ber-EP4). RESULTS: In 13 of 49 patients (26.5 percent), we disclosed tumor cells, mostly located in subcapsular sinuses as single cells or in groups. There was a good correlation between the detection rate and N category, tumor stage, and grading. Moreover, 33 percent of patients in Stage I/II with occult tumor cells (NO+) developed a local relapse and/or distant metastases in contrast to 12 percent of patients without tumor cells (NO−). With a median follow-up of 84 months, we found no difference in disease-free survival between the tumor cell negative and positive groups in Stage I/II patients. CONCLUSION: The results show that occult tumor cells might increase the risk for development of a local tumor relapse and/or distant metastases but do not influence patients' prognoses at all.
Similar content being viewed by others
References
Gusterson B. Are micrometastases clinically relevant? Br J Hosp Med 1992;47:247–8.
Cutait R, Alves VA, Lopes LC,et al. Restaging of colorectal cancer based on the identification of lymph node micrometastases through immunoperoxidase staining of CEA and cytokeratins. Dis Colon Rectum 1991;34:917–20.
Davidson BR, Sams VR, Styles J, Deane C, Boulos PB. Detection of occult nodal metastases in patients with colorectal carcinoma. Cancer 1990;65:967–70.
Greenson JK, Isenhart CE, Rice R, Mojzisik C, Houchens D, Martin EW Jr. Identification of occult micrometastases in pericolic lymph nodes of Dukes' B colorectal cancer patients using monoclonal antibodies against cytokeratin and CC49. Cancer 1994;73:563–9.
Jeffers MD, O'Dowd GM, Mulcahy H, Stagg M, O'Donoghue DP, Toner M. The prognostic significance of immunohistochemically detected lymph node micrometastases in colorectal carcinoma. J Pathol 1994;172:183–7.
Friedman S, Bertin F, Mouriesse H. Importance of tumor cells in axillary node sinus margins (“clandestine metastasis”) discovered by serial sectioning in operable breast carcinoma. Acta Oncol 1988;27:483–7.
International (LUDWIG) Breast Cancer Study Group. Prognostic importance of occult axillary lymph node micrometastases from breast cancers. Lancet 1990;335:1565–8.
Trojani M, De Mascarel I, Bonichon F, Coindre JM, Delsol G. Micrometastases to axillary lymph nodes from carcinoma of breast: detection by immunohistochemistry and prognostic significance. Br J Cancer 1987;55:303–6.
Hermanek P, Scheibe O, Spiessl B, Wagner G, eds. TNM Klassifikation maligner Tumoren. 4. Aufl. Berlin: Springer, 1987.
Cordell JL, Falini B, Erber WN,et al. Immunoenzymatic labeling of monoclonal antibodies using immune complexes of alkaline phosphatase and monoclonal antialkaline phosphatase (APAAP complexes). J Histochem Cytochem 1984;32:219–29.
Garin-Chesa P, Rettig J, Melamed R. Expression of cytokeratin in normal and neoplastic colonic epithelial cells: implications for cellular differentiation and carcinogenesis. Am J Surg Pathol 1986;10:829–35.
Latza U, Niedobitek G, Schwarting R, Nekarda H, Stein H. Ber-EP4: new monoclonal antibody which distinguishes epithelia from mesothelia. J Clin Pathol 1990;43:213–9.
Hermanek P. pTNM and residual tumor classifications: problems of assessment and prognostic significance. World J Surg 1995;19:184–90.
Hermanek P, Sobin LH. Colorectal carcinoma. In: Hermanek P, Gospodarowicz MK, Henson DE, Hutter RV, Sobin LH, eds. Prognostic factors in cancer. Berlin: Springer, 1995:64–79.
Hermanek P, Henson DE, Hutter RV, Sobin LH, eds. UICC TNM Supplement 1993. Berlin: Springer, 1993.
Loy TS, Haege DD. B72.3 immunoreactivity in benign abdominal lymph nodes associated with gastrointestinal disease. Dis Colon Rectum 1995;38:983–7.
Passlick B, Thetter O, Pantel K,et al. Relevance of regional lymphatic tumor cell dissemination in patients with resectable non-small cell lung cancer: results of a prospective study. Chirurg 1995;66:780–6.
Siewert JR, Kestlmeier R, Busch R,et al. Benefits of D2 lymph node dissection for patients with gastric cancer and pN0 and pN1 lymph node metastases. Br J Surg 1996;83:1144–7.
Mori M, Mimori K, Inoue H,et al. Detection of cancer micrometastases in lymph nodes by reverse transcriptasepolymerase chain reaction. Cancer Res 1995;55:3417–20.
Hayashi N, Ito I, Yanagisawa A,et al. Genetic diagnosis of lymph-node metastasis in colorectal cancer. Lancet 1995;345:1257–9.
Arnold MW, Hitchcock CL, Young DC, Burak WE, Bertsch DJ, Martin EW Jr. Intra-abdominal patterns of disease dissemination in colorectal cancer identified using radioimmunoguided surgery. Dis Colon Rectum 1996;39:509–13.
Cote RJ, Houchens DP, Hitchcock CL,et al. Intraoperative detection of occult colon cancer micrometastases using125I-radiolabeled monoclonal antibody CC49. Cancer 1996;77:613–20.
Author information
Authors and Affiliations
About this article
Cite this article
Broll, R., Schauer, V., Schimmelpenning, H. et al. Prognostic relevance of occult tumor cells in lymph nodes of colorectal carcinomas. Dis Colon Rectum 40, 1465–1471 (1997). https://doi.org/10.1007/BF02070713
Issue Date:
DOI: https://doi.org/10.1007/BF02070713