Abstract
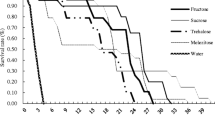
The current year's growth of Douglas fir contains galactose, unusual in that this carbohydrate makes up 78.7% of the total carbohydrate fraction. An agar diet study was undertaken to determine the effects of galactose, other carbohydrates, and terpenes on western spruce budworm larval mortality, growth rate, and adult biomas production. All concentrations of the carbohydrates and terpenes tested, as well as other mineral elements not tested, were typical of the current year's foliage of Douglas fir. In experiment I, the diet containing 5.61% total carbohydrate did not significantly affect larval mortality when compared to the control diet. However, diets containing 9.45% and 15% total carbohydrate concentrations significantly increased larval mortality 64% and 96.1%, respectively, when compared to the control. Also in experiment I, terpenes alone (78.9% morality) and terpenes in combination with 9.45% and 15% total carbohydrates significantly increased larval mortality (97.2% and 100%, respectively) when compared to mortality on the control diet (44%). To determine which carbohydrate was causing the adverse effect, 6% glucose, 6% fructose, and 6% galactose were placed individually and in combination with terpenes in diets in experiment II. The 6% galactose diet significantly increased larval mortality and reduced growth rate when compared to the control, glucose, and fructose diets. Glucose resulted in 16% less larval mortality, significantly enhanced female larval growth rate and pupal weight, but did not affect male larval growth rate and pupal weight, when compared to the control. Fructose resulted in a significant decrease in larval mortality and a general trend of enhanced female and male larval growth rate and pupal weight. Larval mortality on terpenes alone was not significantly different from the control, but terpenes with 6% galactose increased larval mortality and decreased female and male growth rate and pupal weight significantly when compared to glucose-terpene and fructose-terpene diets. No significant interactions were found between carbohydrates and terpenes in either experiment.
Similar content being viewed by others
References
Albert, P.J., Cearly, C., Hanson, F., andParisella, S. 1982. Feeding responses of eastern spruce budworm larvae to sucrose and other carbohydrates.J. Chem. Ecol. 8:233–239.
Altmann, P.L., andDittmer, D.S. (eds.). 1968. Metabolism. Federation of the American Society of Experimental Biology, Bethesda, Maryland, pp. 148–167.
Cates, R.G., andZou J. 1990. Douglas-fir (Pseudotsuga menziesii) in terpene chemistry and its role in budworm (Choristoneura occidentalis Freeman) dynamics, pp. 69–182,in A. Watt, S. Leatyer, M. Hunter, and N. Kidd (eds.). Population Dynamics of Forest Insects. Intercept Ltd., London, England.
Cates, R.G., Henderson, C.B., andRedak, R.A. 1987. Responses of the western spruce budworm to varying levels of nitrogen and terpenes.Oecologia 73:312–316.
Chalupa, V., andFraser, D.A. 1968. Effect of soil and air temperature on soluble sugars and growth of white spruce (Picea glauca) seedlings.Can. J. Bot. 46:65–69.
Clancy, K. 1992. The role of sugars in western spruce budworm nutritional ecology.Ecol. Entomol. 17:189–197.
Clancy, K.M. 1991. Douglas-fir nutrients and terpenes as potential factors influencing western spruce budworm defoliation. Forest Insect Guilds: Patterns of Interaction with Host Trees. USDA Forest Service Gen. Tech. ME-153, pp. 124–134.
Clancy, K.M., Wagner, M.R., andTinus, R.W. 1988. Variations in nutrient levels as a defense: Identifying key nutritional traits of host plants of the western spruce budworm, pp. 203–213,in W.J. Mattson, J. Lerieux, C. Benard-Dagan (eds.), Mechanism of Woody Plant Defenses Against Insects: Search for pattern. Springer-Verlag, New York.
Dixon, A.F.G. 1973. Biology of Aphids. Camelot Press, London.
Harvey, G.T. 1974. Nutritional studies of eastern spruce budworm (Lepidoptera: Tortricidae).Can. Entomol. 106:353–365.
Hedin, P.A. (ed.). 1983. Plant Resistance to Insects. American Chemical Society, Washington, D.C., p. 375.
Heron, R.J. 1965. The role of chemotactic stimuli in the feeding behavior of spruce budworm on white spruce.Can. J. Zool. 43:247–269.
Little, C.H.A. 1970. Seasonal changes in carbohydrate and moisture content in needles of balsam fir (Abies balsamea).Can. J. Bot. 48:2021–2028.
Mattson, W.J., Slocum, S.S., andKoller, C.N. 1983. Spruce budworm (Choristoneura fumiferana) performance in relation to foliar chemistry of its host plants. Forest Defoliator-Host Interactions: A Comparison between Gypsy Moth and Spruce Budworms. U.S. Department of Agriculture, Forest Service., N.E. Forest Experiment Station, Broomall, Pennsylvania, General Technical Report NE-85.
McNeil, S., andSouthwood, T.R.E. 1978. The role of nitrogen in the biochemical aspects of plant and animal coevolution, pp. 77–98,in J. Harborne (ed.). Biochemical Aspects of Plant and Animal Coevolution. Academic Press, New York.
Redak, R.A., andCates, R.G. 1984. Douglas-fir (Pseudotsuga menziesii/spruce budworm (Choristoneura occidentalis) interactions: The effect of nutrition, chemical defenses, tissue phenology, and tree physical parameters on budworm success.Oecologia 2:61–67.
Reese, J.C. 1983. Nutrient-allelochemical interactions in host plant resistance, pp. 231–243,in P. Hedin (ed.). Plant Resistance to Insects. American Chemical Society Symposium Series 208, American Chemical Society, Washington, D.C.
Robertson, J.L. 1979. Rearing the western spruce budworm. USDA Forest Service, Washington, D.C., GPO-1979-699-944.
Rosenthal, G.A., andJanzen, D.H. 1979. Herbivores: Their Interaction with Secondary Plant Metabolites. Academic Press, New York, 718 pp.
SAS Institute, Inc., 1985. SAS Users Guide: Statistics. SAS Institute, Inc., Cary, North Carolina, 956 pp.
Scribner, J.M., andSlansky, F. 1981. The nutritional ecology of immature insects.Annu. Rev. Ecol. Syst. 26:183–211.
Stamp, N.E. 1990. Growth versus molting time of caterpillars as a function of temperature, nutrient, concentration and the phenolic rutin.Oecologia 82:107–113.
White, T.C.R. 1969. An index to measure weather-induced stress of trees associated with outbreaks of psyllids in Australia.Ecology 50:905–909.
White, T.C.R. 1974. A hypothesis to explain outbreaks of looper caterpillars, with special reference to populations ofSelidosema suavis in a population ofPinus radiata in New Zealand.Oecologia 16:279–301.
Zou, J. andCates, R.G. Seasonal variation in foliage constituents of Douglas fir (Pseudotsuga menziesii) and their potential role in Douglas fir resistance and silviculture management.J. Chem. Ecol. Submitted.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zou, J., Cates, R.G. Role of Douglas fir (Pseudotsuga menziesii) carbohydrates in resistance to budworm (Choristoneura occidentalis). J Chem Ecol 20, 395–405 (1994). https://doi.org/10.1007/BF02064446
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02064446