Abstract
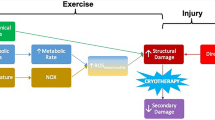
Ischemia reperfusion injury to skeletal muscle may be explained by a cascade of cellular and systemic events initiated by an ischemic period followed by reperfusion. During the period of ischemia there is a gradual reduction of intracellular energy stores. Adenosine triphosphate is gradually depleted despite the buffering effect of creatine phosphate which is present in large stores in muscles. As well, glycogen stores are depleted with resultant production of small amounts of energy and large accumulations of lactate. Upon reperfusion there is a reactive hyperemia, resulting in an overall increase in muscle blood flow, despite the fact that areas may continue to be underperfused. Results of this blood flow are mixed with the beneficial effects of removing metabolic by-products and supplying exogenous substrates and oxygen. However, this blood flow also causes harmful effects by washing out necessary precursors for adenine nucleotide resynthesis. Production of oxygen free radicals occurs with resultant membrane lipid peroxidation, and calcium influx occurs upon reoxygenation with resultant disruption of oxidative rephosphorylation in the mitochondria. The sequestration of white blood cells in the muscle due to up regulation of both neutrophil receptors and endothelial leukocyte adhesion molecules results in a prolongation of the reperfusion injury. This subsequently results in damage to remote organs, including lung, heart, and kidneys. The future for therapeutic interventions aimed at reducing this injury lie mostly in the ability to modulate the reperfusion effect.
Similar content being viewed by others
References
RUDERMAN NB, GOODMAN MN, BERGER M, et al. Effect of starvation on muscle glucose metabolism: studies with the isolated perfused rat hindquarter.Fed Proc 1977;36:171–176.
HALJAMAE H, ENGER E. Human skeletal muscle energy metabolism during and after complete tourniquet ischemia.Ann Surg 1975;182:9–14.
HULTMAN E. Muscle glycogen in man determined in needle biopsy specimens. Method and normal values.Scand J Clin Lab Invest 1967;19:209–217.
HARRIS K, WALKER P, MICKLE D, et al. Metabolic response of skeletal muscle to ischemia.Am J Physiol 1986;(250)19:H213-H220.
LARSSON J, HULTMAN E. The effect of long term arterial occlusion on energy metabolism of the human quadriceps muscle.Scand J Clin Lab Invest 1979;39:257–264.
ATKINSON DE. The energy charge of the adenylate pool as a regulatory parameter. Interaction with feedback modifiers.Biochemistry 1968;7:4030–4034.
LINDSAY T, LIAUW K, ROMASCHIN A, et al. The effect of ischemia/reperfusion on adenine nucleotide metabolism and xanthine oxidase production in skeletal muscle.J Vasc Surg 1990;12(1):8–15.
TULLSON P, J-ADLER H, HOOD D, et al. De novo synthesis of adenine nucleotides in different skeletal muscle fiber types.Am Physiol Soc 1988;C271–C277.
REIMER KA, JENNINGS RB, TATUM AH. Pathobiology of acute myocardial ischemia; metabolic, functional and ultrastructural studies.Am J Cardiol 1983;52:72A-81A.
FORREST I, LINDSAY T, ROMASCHIN A, et al. The rate and distribution of muscle blood flow after prolonged ischemia.J Vasc Surg 1989;10(1):83–88.
ADAMS P, ADAMS F, BELL P, et al. Impaired renal blood flow in acute renal failure.Kidney Int 1980;16:68–76.
ENGLER R, DAHLGREN M, PETERSON M, et al. Accumulation of polymorphonuclear leukocytes during 3h experimental myocardial ischemia.Am J Physiol 1986;251: H93-H100.
KORTHIUS R, GRANGER D, FORONSKY M, et al. The role of oxygen derived free radicals in ischemia induced increases in canine skeletal muscle vascular permeability.Circ Res 1985;57:599–609.
LINDSAY T, WALKER P, MICKLE D, et al. Measurement of hydroxy conjugated dienes after ischemia/reperfusion in canine skeletal muscle.Am J Physiol 1988;254(23): H578-H583.
MC CORD J. Oxygen derived free radicals in post ischemic tissue injury.N Engl J Med 1985;312:159–163.
CRAKE T, POOLE-WILSON P. Evidence that calcium influx on reoxygenation is not due to cell membrane disruption in isolated rabbit heart.J Mol Cell Cardiol 1986;18:31.
SMITH A, HAYES G, ROMASCHIN A, et al. The role of extracellular calcium in ischemia/reperfusion injury in skeletal muscle.J Surg Res 1990;49:1–4.
RUBIN B, SMITH A, LIAUW K, et al. Complement activation and white cell sequestration in post ischemic skeletal muscle.Am J Physiol 1990;259(28):H525-H531.
RUBIN B, SMITH A, LIAUW K, et al. Skeletal muscle ischemia stimulates an immune system mediated injury.Surg For 1989;Vol XL:297–300.
SIMPSON P, FANTONE J, MICKELSON J, et al. Identification of a time window for therapy to reduce experimental canine myocardial injury; suppression of neutrophil activation during 72 hours of reperfusion.Circ Res 1988;65:1070–1079.
ENGLER R, SCHMID-SCHONBEIN G, PAVALEC R. Leukocyte capillary plugging in myocardial ischemia and reperfusion in the dog.Am J Pathol 1983;111:98–111.
UNAS S, SHANLEY P, WHITTENBERG D, et al. Neutrophils accentuate ischemia/reperfusion injury in isolated perfused rat kidneys.Am J Physiol 1988;255(24):F728-F735.
HAIMOVICI H. Muscular, renal and metabolic complications of acute arterial occlusions; myonephropathic metabolic syndrome.Surgery 1979;85:461–468.
LEWIS R, RICE J, PATTON M, et al. Renal ischemic injury in the dog; characterizations and effect of various pharmacologic agents.J Lab Clin Med 1984;104:470–479.
GOLDMAN G, WELBOURN R, KLAUSNER J, et al. Ischemia activates neutrophils but inhibits their local and remote diapedesis.Ann Surg 1990;211: 196–201.
RUBIN B, TITTLEY J, CHANG G, et al. A clinically applicable method for long term salvage of post ischemic skeletal muscle.J Vasc Surg 1991;13:58–68.
CARDEN DL, SMITH JK, and KORTHUIS RJ. Neutrophil-mediated microvascular dysfunction in postischemic canine skeletal muscle.Circ Res 1990;66:1436–1444.
Author information
Authors and Affiliations
About this article
Cite this article
Walker, P.M. Ischemia/reperfusion injury in skeletal muscle. Annals of Vascular Surgery 5, 399–402 (1991). https://doi.org/10.1007/BF02015307
Issue Date:
DOI: https://doi.org/10.1007/BF02015307