Abstract
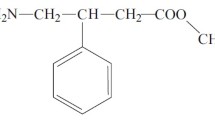
A-56268 is a new macrolide which is generally two-fold more potent than erythromycin. A new bioassay is described in which plasma samples are extracted with acetonitrile prior to bioassay. The concentration range for the assay is between 0.05–4.0 µg/ml, and the concentrations measured are within 6% of those measured by high-power liquid chromatography. An active metabolite which is as active as erythromycin was identified in the plasma. The plasma half-life and area under the plasma curve values of A-56268, as determined by bioassay, were significantly greater than those of erythromycin.
Similar content being viewed by others
References
Fernandes, P.B., Bailer, R., Swanson, R., Hanson, C.W., McDonald, E., Ramer, N., Hardy, D., Shipkowitz, N., Bower, R.R., Gade, E. In vitro and in vivo evaluation of A-56268 (TE-031): a new macrolide. Antimicrobial Agents and Chemotherapy 1986, 30: 865–873.
Fernandes, P.B. The macrolide revival: 35 years after erythromycin. Antimicrobic Newsletter 1987, 4: 25–34.
Code of Federal Regulations. Food and Drugs Microbiological assay methods. Volume 21, Section 436.100. US Government Printing Office, Washington, DC, 1986.
National Committee for Clinical Laboratory Standards: Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. Approved standard. M7-A, Villanova, PA, 1985.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fernandes, P.B., Ramer, N., Rode, R.A. et al. Bioassay for A-56268 (TE-031) and identification of its major metabolite, 14-hydroxy-6-O-methyl erythromycin. Eur. J. Clin. Microbiol. Infect. Dis. 7, 73–76 (1988). https://doi.org/10.1007/BF01962181
Issue Date:
DOI: https://doi.org/10.1007/BF01962181