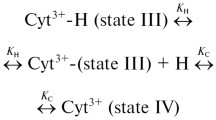Abstract
The effect of glycerol on the structure and redox properties of horse heart cytochrome c was investigated by absorption spectroscopy, circular dichroism, and dc cyclic voltammetry techniques. The results show that the organic solvent increases the α-helix structure of the protein and induces slight changes at the active-site environment; however, the overall tertiary structure does not appear to be significantly perturbed. Glycerol stabilizes cytochrome c, the free energy of denaturation (ΔG 0) being approximately 0.7 kcal/mol larger than that determined in phosphate buffer under the same conditions, and influences the heterogeneous electron transfer kinetics at a chemically modified gold electrode; on the other hand, the redox potential of the protein is unaltered. On the whole, the results obtained indicate that glycerol acts as a suitable stabilizing agent of cytochrome c, which is of interest for application in biotechnology; the organic solvent does not alter the tertiary structure significantly or the redox properties of the protein. This has to be interpreted not only in terms of the glycerol-induced solvent ordering around the protein surface, but also as due to the specific features of the protein matrix.
Similar content being viewed by others
References
Bard, A. J., and Faulkner, L. R. (1980). InElectrochemical Methods. Fundamental and Applications, Wiley, New York, Chapter 6.
Barteri, M., Gaudiano, M. C., and Santucci, R. (1996).Biochim. Biophys. Acta,1295, 51–58.
Bond, A. M. (1980). InModern Polarographyc Methods in Analytical Chemistry, Marcel Dekker, New York.
Bushnell, G. W., Louie, G. V., and Brayer, G. D. (1990).J. Mol. Biol. 214, 585–595.
Colombo, M. F., Rau, D. C., and Parsegian, V. A. (1992).Science 256, 655–659.
Cupane, A., Leone, E., and Vitrano, E. (1993).Eur. Biophys. J. 21, 385–390.
Dickerson, R. E., and Timkovich, R. (1975).The Enzymes, Vol. XI,Cytochromes C (Boyer, P., ed.). Academic Press, New York, pp. 397–492.
Dill, K. A. (1990).Biochemistry 29, 7133–7155.
Eddows, M. J., and Hill, H. A. O. (1979).J. Am. Chem. Soc. 101, 4461–4464.
Gekko, K., and Timasheff, S. M. (1981).Biochemistry 20, 4667–4686.
Greene, R. F., and Pace, C. N. (1974).J. Biol. Chem. 249, 5388–5393.
Greenfield, N., and Fasman, G. D. (1969).Biochemistry 8, 4108–4116.
Herskovits, T. T., Gadegbeku, B., and Jaillet, H. (1970).J. Biol. Chem. 245, 2588–2598.
Hirst, J. D., and Brooks III, C. L. (1994).J. Mol. Biol. 243, 173–178.
Hobbs, J. D., and Shelnutt, J. A. (1995).J. Protein Chem. 14, 19–25.
Kazandjian, R. Z., and Klibanov, A. M. (1985).J. Am. Chem. Soc. 107, 5448–5452.
Kim, P. S., and Baldwin, R. L. (1990).Annu. Rev. Biochem. 59, 631–660.
Lo, T. P., Komar-Panicucci, S., Sherman, F., McLendon, G., and Brayer, G. D. (1995).Biochemistry 34, 5259–5268.
Louie, G. V., and Brayer, G. D. (1989).J. Mol. Biol. 210, 313–322.
Louie, G. V., Pielak, G. J., Smith, M., and Brayer, G. D. (1988).Biochemistry 27, 7870–7876.
Mabrouk, P. A. (1995).J. Am. Chem. Soc. 117, 2141–2146.
Nedjar-Arroume, N., Guillochon, D., Piot, J. M., and Thomas, D. (1991).Biotechnol. Appl. Biochem. 13, 303–314.
Nedjar-Arroume, N., Castellano, A., Piot, J. M., and Guillochon, D. (1993).Biotechnol. Appl. Biochem. 18, 25–35.
Nicholson, R. S. (1965).Anal. Chem. 37, 1315–1355.
Nicholson, R. S., and Shain, I. (1964).Anal. Chem. 36, 706–723.
Pace, C. N. (1975).CRC Crit. Rev. Biochem. 3, 1–45.
Rand, R. P. (1992).Science 256, 618.
Rose, G. D., Geselowitz, A. R., Lesser, G. J., Lee, R. H., and Zehfus, M. (1985).Science 229, 834–838.
Sage, J. T., Schomacker, K. T., and Champion, P. M. (1995).J. Phys. Chem. 99, 3394–3405.
Schejter, A., and Aviram, I. (1970).J. Biol. Chem. 245, 1552–1557.
Schellman, J. A. (1990).Biophys. Chem. 37, 121–140.
Senn, H., and Wuthrich, K. (1985).Q. Rev. Biophys. 18, 111–134.
Srajer, V., Schomacker, K. T., and Champion, P. M. (1986).Phys. Rev. Lett. 57, 1267–1305.
Staniforth, R. A., Burston, S. G., Smith, C. J., Jackson, G. S., Badcoe, I. G., Atkinson, T., Holbrook, J. J., and Clarke, A. R. (1993).Biochemistry 32, 3842–3851.
Takano, T., and Dickerson, R. E. (1981).J. Mol. Biol. 153, 79–115.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
De Sanctis, G., Maranesi, A., Ferri, T. et al. Influence of glycerol on the structure and redox properties of horse heart cytochrome c. A circular dichroism and electrochemical study. J Protein Chem 15, 599–606 (1996). https://doi.org/10.1007/BF01886742
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01886742