Summary
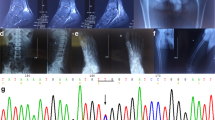
Extracellular-superoxide dismutase (EC-SOD) is one of the SOD isozymes mainly distributed in the extracellular fluid. In the vascular system, it is located on the endothelial cell surface according to studies on the heparin binding capacity. By measurement of serum EC-SOD levels of Japanese in healthy persons (n=103) and hemodialysis patients (n=150), 7 healthy subjects and 24 hemodialysis patients were classified into group II associated with high EC-SOD levels. By molecular analysis of the EC-SOD coding region from the group II individuals in Sweden, a single nucleotide substitution of G to C generating an amino acid change of arginine to glycine has been identified in the region associated with the heparin affinity of the enzyme. The same mutation was detected in the Japanese as a homozygote in both alleles of 2 hemodialysis patients and as a heterozygote in one allele of all the healthy group II individuals and 17 hemodialysis patients. The amino acid substitution may result in the decrease of the heparin affinity which is favorable for the existence of EC-SOD in the serum.
Similar content being viewed by others
Article PDF
References
Adachi, T Ohta H, Yamada H, Futenma A, Kato K, Hirano K (1992a): Quantitative analysis of extracellular-superoxide dismutase in serum and urine by ELISA with monoclonal antibody. Clin Chim Acta212: 89–102
Adachi T, Kodera T, Ohta H, Hayashi K, Hirano K (1992b): The heparin binding site of human extracellular-superoxide dismutase. Arch Biochem Biophys297: 155–161
Adachi T, Nakamura M, Yamada H, Kitano M, Futenma A, Kato K, Hirano K (1993): Pedigree of serum extracellular-superoxide dismutase level. Clin Chim Acta223: 185–187
Adachi T, Nakamura M, Yamada H, Futenma A, Kato K, Hirano K (1994): Quantitative and qualitative change of extracellular-superoxide dismutase in patients with various disease. Clin Chim Acta229: 123–131
Adachi T, Marklund SL (1989): Interactions between human extracellular superoxide dismutase C and sulfated polysaccharides. J Biol Chem264: 8537–8541
Hjalmarsson K, Marklund SL, Engström A, Edulund T (1987): Isolation and sequence of complementary DNA encoding human extracellular superoxide dismutase. Proc Natl Acad Sci USA84: 6340–6344
Inoue M, Watanabe N, Utsumi T, Sasaki J (1991a): Targeting SOD by gene and protein engineering and inhibition of free radical injury. Free Radical Res Commun12: 391–399
Inoue M, Watanabe N, Matsuno K, Sasaki J, Tanaka Y, Hatanaka H, Amachi T (1991b): Expression of a hybrid Cu,Zn-type superoxide dismutase which has high affinity for heparin-like proteoglycans on vascular endothelial cells. J Biol Chem266: 1409–16414
Karlsson K, Lindahl U, Marklund SL (1988): Binding of human extracellular superoxide dismutase C to sulphated glycosaminoglycans. Biochem J256: 29–33
Karlsson K, Edlund A, Sandström J, Marklund SL (1993): Proteolytic modification of the heparin-binding affinity of extracellular superoxide dismutase. Biochem J290: 623–626
Marklund SL (1984a): Extracellular superoxide dismutase and other superoxide dismutase isozymes in tissues from nine mammalian species. Biochem J222: 649–655
Marklund SL (1984b): Extracellular superoxide dismutase in human tissues and human cell lines. J Clin Invest74: 1398–1403
Marklunfi SL (1990): Expression of extracellular superoxide dismutase by human cell lines. Biochem J266: 213–219
Sandström J, Nilsson, P, Karlsson K, Marklund SL (1994): 10-fold increase in human plasma extracellular superoxide dismutase content caused by a mutation in heparin-binding domain. J Biol Chem269: 19163–19166
Sandström J, Carlsson L, Marklund SL, Edlund T (1992): The heparin-binding domain of extracellular superoxide dismutase C and formation of variants with reduced heparin affinity. J Biol Chem267: 18205–18209
Shah SV (1989): Role of reactive oxygen metabolites in experimental glomerular disease. Kidney Int35: 1093–1106
Oyanagui Y, Sato S, Inoue M (1991): Inhibition of carrageenan-induced paw edema by superoxide dismutase that binds to heparin sulfates on vascular endothelial cells. Biochem Pharmacol42: 991–995
Yamada Y, Goto H, Ogasawara N (1993): A point mutation responsible for hjman erythrocyte AMP deaminase deficiency. Hum Mol Genet3: 331–334
Yamada Y, Goto H, Suzumori K, Adachi R, Ogasawara N (1992): Molecular analysis of five independent Japanese mutant genes responsible for hypoxanthine guanine phosphoribosyltransferase (HPRT) deficiency. Hum Genet90: 379–384
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yamada, H., Yamada, Y., Adachi, T. et al. Molecular analysis of extracellular-superoxide dismutase gene associated with high level in serum. Jap J Human Genet 40, 177–184 (1995). https://doi.org/10.1007/BF01883574
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01883574
Key Words
This article is cited by
-
Purple grape juice improves performance of recreational runners, but the effect is genotype dependent: a double blind, randomized, controlled trial
Genes & Nutrition (2022)
-
Association among extracellular superoxide dismutase genotype, plasma concentration, and comorbidity in the very old and centenarians
Scientific Reports (2021)
-
Anti-oxidative effects of superoxide dismutase 3 on inflammatory diseases
Journal of Molecular Medicine (2020)
-
Polymorphic Variants of Extracellular Superoxide Dismutase Gene in a Romanian Population with Atheroma
Biochemical Genetics (2008)
-
Polymorphisms in the Mn-SOD and EC-SOD Genes and Their Relationship to Diabetic Neuropathy in Type 1 Diabetes Mellitus
BMC Medical Genetics (2001)