Summary
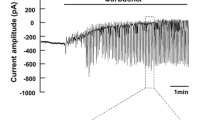
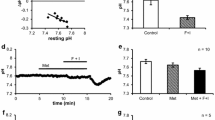
In the isolated, superfused mouse lacrimal gland, intracellular Na+ activities (aNa i ) of the acinar cells were directly measured with double-barreled Na+-selective microelectrodes. In the nonstimulated conditionaNa i was 6.5±0.5 mM and membrane potential (V m ) was −38.9±0.4 mV. Addition of 1 mM ouabain or superfusion with a K+-free solution slightly depolarized the membrane and caused a gradual increase inaNa i . Stimulation with acetylcholine (ACh, 1 μM) caused a membrane hyperpolarization by about 20 mV and an increase inaNa i by about 9 mM in 5 min. The presence of amiloride (0.1 mM) reduced the ACh-induced increase inaNa i by approximately 50%, without affectingV m and input resistance in both nonstimulated and ACh-stimulated conditions. Acid loading the acinar cells by an addition/withdrawal of 20 mM NH4Cl or by replacement of Tris+-buffer saline solution with HCO −3 /CO2-buffered solution increasedaNa i by a few mM. Superfusion with a Cl−-free NO −3 solution or 1 mM furosemide or 0.5 mM bumetanide-containing solution had little effect on the restingaNa i levels, however, it reduced the ACh-induced increase inaNa i by about 30%. Elimination of metabolite anions (glutamate, fumarate and pyruvate) from the superfusate reduced both the restingaNa i and the ACh-induced increase inaNa i .
The present results suggest the presence of multiple Na+ entry mechanisms activated by ACh, namely, Na+/H+ exchange, Na-K-Cl cotransport and organic substrate-coupled Na+ transport mechanisms.
Similar content being viewed by others
References
Alexander, J.H., Lennep, E.W. van, Young, J.A. 1972. Water and electrolyte secretion by the exorbital lacrimal gland of the rat studied by micropuncture and catheterization techniques.Pfluegers Arch. 337:299–309
Aronson, P.S., Nee, J., Suhm, M.A. 1982. Modifier role of internal H+ in activating Na+-H+ exchanger in renal microvillus membrane vesicles.Nature (London) 299:161–163
Brown, H.M., Owen, J.D. 1979. Micro ion-selective electrodes for intracellular ions.Ion-Selective Electrode Rev. 1:145–186
Cassola, A.C., Mollenhauer, M., Fromter, E. 1983. The intracellular chloride activity of rat kidney proximal tubular cells.Pfluegers Arch. 399:259–265
Dartt, D.A., Moller, M., Poulsen, J.H. 1981. Lacrimal gland electrolyte and water secretion in the rabbit: Localization and role of (Na++K+)-activated ATPase.J. Physiol. (London) 321:557–569
Dufresne, M., Bastie, M.J., Vaysse, N., Creach, Y., Hollande, E., Ribet, A. 1985. The amiloride sensitive Na+/H+ antiport in guinea pig pancreatic acini: Characterization and stimulation by celrulein.FEBS Lett. 187:126–130
Hellmessen, W., Christian, A.L., Fasold, H., Schulz, I. 1985. Coupled Na+-H+ exchange in isolated acinar cells from rat exocrine pancreas.Am. J. Physiol. 249:G125-G136
Henniger, R.A., Schulte, B.A., Spicer, S.S. 1983. Immunolocalization of carbonic anhydrase isozymes in rat and mouse salivary and exorbital lacrimal glands.Anat. Rec. 207:605–614
Herzog, V., Sies, H., Miller, F. 1976. Exocytosis in secretory cells of rat lacrimal gland. Peroxidase release from lobules and isolated cells upon cholinergic stimulation.J. Cell Biol. 70:692–706
Iwatsuki, N., Petersen, O.H. 1978. Intracellular Ca2+ injection causes membrane hyperpolarization and conductance increase in lacrimal acinar cells.Pfluegers Arch. 377:185–187
Kinsella, J.L., Aronson, P.S. 1980. Properties of the Na+-H+ exchanger in renal microvillus membrane vesicles.Am. J. Physiol. 238:F461-F469
Kuijpers, G.A.J., Depont, J.J.H.H.M., Nooy, I.G.P. van, Fleuren-Jakobs, A.M.M., Bonting, S.L., Rodrigues De Miranda, J.F. 1984. Amiloride is a cholinergic antagonist in the rabbit pancreas.Biochim. Biophys. Acta 804:237–244
Marty, A., Tan, Y.P., Trautmann, A. 1984. Three types of calcium-dependent channel in rat lacrimal glands.J. Physiol. (London) 357:293–325
Mircheff, A.K., Lu, C., Conteas, C.N. 1983. Resolution of apical and basal-lateral membrane populations from rat exorbital gland.Am. J. Physiol. 245:G661-G667
O'Doherty, J., Stark, R.J. 1983. A transcellular route for Nacoupled Cl transport in secreting pancreatic acinar cells.Am. J. Physiol. 245:G499-G503
Parod, R.J., Leslie, B.A., Putney, J.W., Jr. 1980. Muscarinic and α-adrenergic stimulation of Na and Ca uptake by dispersed lacrimal cells.Am. J. Physiol. 239:G99-G105
Parod, R.J., Putney, J.W., Jr. 1980. Stimulus-permeability coupling in rat lacrimal gland.Am. J. Physiol. 239:G106-G113
Petersen, O.H., Maruyama, Y. 1984. Calcium-activated potassium channels and their role in secretion.Nature (London) 307:693–695
Saito, Y., Ozawa, T., Hayashi, H., Nishiyama, A. 1985a. Acetylcholine-induced change in intracellular Cl− activity of the mouse lacrimal acinar cells.Pfluegers Arch. 405:108–111
Saito, Y., Ozawa, T., Hayashi, H., Nishiyama, A. 1985b. Cl and K activities in the mouse lacrimal acinar cells and the effect of secretagogues.J. Physiol. Soc. Jpn. 47:389
Saito, Y., Ozawa, T., Hayashi, H., Nishiyama, A. 1987. The effect of acetylcholine on chloride transport across the mouse lacrimal gland acinar cell membranes.Pfluegers Arch. (in press)
Saito, Y., Ozawa, T., Nishiyama, A. 1986a. Multiple Na entry mechanisms in the mouse lacrimal gland acinar cells as studied with Na-selective microelectrode method.In: Gastrointestinal Secretion Symposium. July, 1986, Calgary, Alberta, Canada. The proceedings. (in press)
Saito, Y., Ozawa, T., Nishiyama, A. 1986b. Transcellular chloride transport by acinar cells of the mouse lacrimal gland.Proc. Int. Union Physiol. Sci. 16:p480
Seow, K.T.F., Lingard, J.M., Young, J.A. 1986. Anionic basis of fluid secretion by rat pancreatic acini in vitro.Am. J. Physiol. 250:G140-G148
Soltoff, S.P., Mandel, L.J. 1983. Amiloride directly inhibits the Na, K-ATPase activity of rabbit kidney proximal tubules.Science 220:957–959
Steiner, R.A., Oehme, M., Ammann, D., Simon, W. 1979. Neutral carrier sodium ion-selective microelectrode for intracellular studies.Anal. Chem. 51:351–353
Suzuki, K., Petersen, O.H. 1985. The effect of Na and Cl removal and of loop diuretics on acetylcholine-evoked membrane potential changes in mouse lacrimal acinar cells.Q. J. Exp. Physiol. 70:437–445
Thomas, R.C. 1974. Intracellular pH of snail neurones measured with a new pH-sensitive glass micro-electrode.J. Physiol. (London) 238:159–180
Vogh, B.P., Langham, M.R., Jr. 1981. The effect of furosemide and bumetanide on cerebrospinal fluid formation.Brain Res. 221:171–183
Wright, E.M. 1984. Electrophysiology of plasma membrane vesicles.Am. J. Physiol. 246:F363-F372
Yoshitomi, K., Fromter, E. 1984. Cell pH of rat proximal tubule in vivo and the conductive nature of peritubular HCO −3 (OH−) exit.Pfluegers Arch. 402:300–305
Zeuthen, T. 1980. How to make and use double-barreled ionselective microelectrodes.Curr. Topics Membr. Transp. 13:31–47
Zeuthen, T. 1983. Ion activities in the lateral intercellular spaces of gallbladder epithelium transporting at low external osmolarities.J. Membrane Biol. 76:113–122
Zeuthen, T., Machen, T. 1984. HCO −3 /CO2 stimulates Na+/H+ and Cl−/HCO −3 exchange inNecturus gallbladder.In: Hydrogen Ion Transport in Epithelia. J.G. Forte, D.G. Warnock, and F.C. Rector, Jr., editors. pp. 97–108. Wiley, New York
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Saito, Y., Ozawa, T. & Nishiyama, A. Acetylcholine-Induced Na+ influx in the mouse lacrimal gland acinar cells: Demonstration of multiple Na+ transport mechanisms by intracellular Na+ activity measurements. J. Membrain Biol. 98, 135–144 (1987). https://doi.org/10.1007/BF01872126
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01872126