Summary
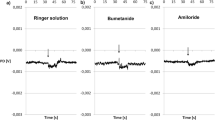
Transepithelial Li+ influx was studied in the isolated epithelium from abdominal skin ofRana catesbeiana. With Na+-Ringer's as inside medium and Li+-Ringer's as outside medium, the Li+ influx across the epithelium was 15.6 μA/cm2. This influx was considerably reduced by removal of either Na+ or K+ from the inside bath or by the addition of ouabain or amiloride. Epithelial K+ or Na+ concentration was respectively lower in epithelia bathed in K+-free Ringer's or Na+-free Ringer's. In conditions of negligible Na+ transport, a 20mm Li+ gradient (out→in) produced across the short-circuited epithelium a Li+ influx of 11.8 μA/cm2 and a mean short-circuit current of 10.2 μA/cm2. The same Li+ gradient in the opposite direction produced a Li+ outflux of only 1.9 μA/cm2. With equal Li+ concentration (10.3 and 20.6mm) on both sides of the epithelium, plus Na+ in the inside solution only, a stable Li+-dependent short-circuit current was observed. Net Li+ movement (out→in) was also indirectly determined in the presence of an opposing Li+ gradient. Although Li+ does not substitute for Na+ as an activator of the (Na++K+)-ATPase from frog skin epithelium, Li+ influx appears to be related to Na+−K+ pump activity. It is proposed that the permeability of the “outer barrier” to Na+ and Li+ is regulated by the electrical gradient produced by electrogenic Na+−K+ pumps located in the membrane of the deeper epithelial cells.
Similar content being viewed by others
References
Aceves, J., Erlij, D. 1971. Sodium transport across the isolated epithelium of the frog skin.J. Physiol. 212:195
Biber, T.U.L., Chez, R.A., Curran, P.E. 1966. Na transport across frog skin at low external Na concentrations.J. Gen. Physiol. 49:1161
Biber, T.U.L., Cruz, L.J. 1973. Effect of antidiuretic hormone on sodium uptake across outer surface of frog skin.Amer. J. Physiol. 225:912
Biber, T.U.L., Curran, P.F. 1970. Direct measurement of uptake of sodium at the outer surface of the frog skin.J. Gen. Physiol. 56:83
Bonting, S.L., Canady, M.R. 1964. Na−K activated adenosine triphosphatase and sodium transport in toad bladder.Amer. J. Physiol. 207:1005
Bonting, S.L., Caravaggio, L.L., Hawkins, N.M. 1962. Studies on sodium-potassium-activated adenosinetriphosphatase. IV. Correlation with cation transport sensitive to cardiac glycosides.Arch. Biochem. Biophys. 98:413
Candia, O.A. 1970. The hyperpolarizing region of the current-voltage curve in frog skin.Biophys. J. 10:323
Candia, O.A. 1972. Ouabain and sodium effects on chloride fluxes across the isolated bullfrog cornea.Amer. J. Physiol. 223:1053
Candia, O.A., Chiarandini, D.J. 1973. Transport of lithium and rectification by frog skin.Biochim. Biophys. Acta 307:578
Candia, O.A., Reinach, P. 1975. The Na pool and Na fluxes across inner and outer barriers of frog skin epithelium.Biophys. J. 15:228a (#2 Abstract)
Candia, O.A., Zadunaisky, J.A. 1972. Potassium flux and sodium transport in the isolated frog skin.Biochim. Biophys. Acta 225:517
Cereijido, M., Herrera, F.C., Flanigan, W.J., Curran, P.F. 1964. The influence of Na concentration on Na transport across frog skin.J. Gen. Physiol. 47:879
Erlij, D., Smith, M.W. 1973. Sodium uptake by frog skin and its modification by inhibitors of transepithelial sodium transport.J. Physiol. 228:221
Finkelstein, A. 1961. Lithium-induced oscillations of potential and resistance in isolated frog skin.J. Gen. Physiol. 44:1165
Kawada, J., Taylor, R.E., Jr., Barker, S.B. 1969. Measurement of Na−K-ATPase in the separated epidermis ofRana catesbeiana frogs and tadpoles.Comp. Biochem. Physiol. 30:965
Koefoed-Johnsen, V. 1957. The effect ofg-Strophanthin (ouabain) on the active transport of sodium through the isolated frog skin.Acta Physiol. Scand. 42 (Suppl. 145):87
Koefoed-Johnsen, V., Ussing, H.H. 1958. The nature of the frog skin potential.Acta Physiol. Scand. 42:298
Leblanc, G. 1972. The mechanism of lithium accumulation in the isolated frog skin epithelium.Pflugers Arch. 337:1
Moreno, J.H., Reisin, I.L., Rodriquez Boulan, E., Rotunno, C.A., Cereijido, M. 1973. Barriers to sodium movement across frog skin.J. Membrane Biol. 11:99
Rabito, C.A., Rodriquez Boulan, E., Cereijido, M. 1973. Effect of the composition of the inner bathing solution on transport properties of the frog skin.Biochim. Biophys. Acta 311:630
Rajerison, R.M., Montegut, M., Jard, S., Morel, F. 1972. The isolated frog skin epithelium: Permeability characteristics and responsiveness to oxytocin, cyclic AMP and theophylline.Pflugers Arch. 332:302
Rotunno, C.A., Vilallonga, F.A., Fernandez, M., Cereijido, J. 1970. The penetration of sodium into the epithelium of the frog skin.J. Gen. Physiol. 55:716
Rotunno, C.A., Zylber, E.A., Cereijido, M. 1973. Ion and water balance in the epithelium of the abdominal skin of the frogLeptodactylus ocellatus.J. Membrane Biol. 13:217
Siegel, G.J., Tormay, A., Candia, O.A. 1975. Microsomal sodium-potassium-activated adenosinetriphosphatase from frog skin epithelium: Cation activation and some effects of inhibitors.Biochim. Biophys. Acta 389:557
Ussing, H. 1965. Transport of electrolytes and water across epithelia.Harvey Lect. 59:1
Zerahn, K. 1969. Nature and localization of the sodium pool during active transport in the isolated frog skin.Acta Physiol. Scand. 77:272
Zylber, E.A., Rotunno, C.A., Cereijido, M. 1973. Ion and water balance in isolated epithelial cells of the abdominal skin of the frogLeptodactylus ocellatus.J. Membrane Biol. 13:199
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Reinach, P.S., Candia, O.A. & Siegel, G.J. Lithium transport across isolated frog skin epithelium. J. Membrain Biol. 25, 75–92 (1975). https://doi.org/10.1007/BF01868569
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01868569