Abstract
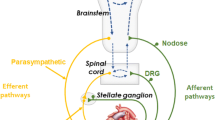
Disturbed autonomic nervous ‘balance’ of the sympathetic nervous and vagal outflows to the heart potentiates the experimental development of ventricular arrhythmias in laboratory animals. For some time the best evidence for the occurrence of a similar phenomemon in humans was provided by the long QT interval syndrome, sufferers of which are very prone to develop serious ventricular arrhythmias and in whom evidence exists of abnormal anatomy and function of the cardiac sympathetic nerves. Recently the case for disturbed autonomic function causing clinical arrhythmias has become more broadly based. Reduced baroreflex sensitivity after myocardial infarction, and low heart rate variability, both of which rest largely on vagal underactivity, have been shown to be associated with substantially increased risk of subsequent sudden death. A second observation is that patients having recovered from unexpected ventricular tachycardia or ventricular fibrillation have markedly increased cardiac sympathetic activity compared with appropriate reference groups, based on measurements of the rate of spillover of the sympathetic neurotransmitter, noradrenaline, from the heart to plasma. These clinical findings support a role for cardiac autonomic dysfunction, specifically sympathetic activation and vagal withdrawal, in arrhythmogenesis. These observations are timely, given the recent demonstration that most conventional anti-arrythmics are of little benefit in preventing sudden death. A reappraisal of the anti-arrhythmic activity of β-adrenergic blocking drugs, and evaluation of potential benefits of other pharmacological and non-pharmacological means of favourably altering cardiac autonomic function is now needed.
Similar content being viewed by others
References
Engel GL. Sudden and rapid death during psychological stress: folklore or folk wisdom?Ann Intern Med 1971;74: 771–782.
Brodsky MA, Sato DA, Iseri LT, Wolff LJ, Allen BJ. Ventricular tachyarrhythmia associated with psychological stress: the role of the sympathetic nervous system.JAMA 1987;257: 2064–2067.
Randall WC, Armour JA. Gross and microscopic anatomy of the cardiac innervation. In: Randall WC, ed.Neural Regulation of the Heart. Chapter 2. New York: Oxford University Press, 1977; 13–41.
Levy MN, Martin PJ. Neural control of the heart. In: Berne RM, Sperelakis N, Geiger SR, eds.Handbook of Physiology, Section 2, The Cardiovascular System, Volume 1, The Heart, Chapter 16. Baltimore: Williams and Wilkins, 1979; 581–620.
Levy MN. Sympathetic—parasympathetic interactions in the heart.Circ Res 1971;29: 437–445.
Lown B, Verrier RL. Neural activity and ventricular fibrillation.N Engl J Med 1976;294: 1165–1170.
Zipes DP. The long QT interval syndrome. A Rosetta stone for sympathetic related ventricular tachyarrhythmias.Circulation 1991;84: 1414–1419.
Rosen KM, Bauernfeind RA. Torsades de points. In: Petersdorf RG, Adams RD, Braunwald E, Isselbacher KJ, Martin JB, Wilson JD, eds.Harrison's Principles of Internal Medicine, Edition 10, Chapter 255. New York: McGraw-Hill, 1983: 1381.
Schwartz PJ, La Rovere MT, Vanoli E. Autonomic nervous system and sudden cardiac death: Experimental basis and clinical observations for post-myocardial infarction risk stratification.Circulation 1992;85 (suppl): 177–191.
Keating M, Atkinson D, Dunn C, Timothy K, Vincent GM, Leppert M. Linkage of a cardiac arrhythmia, the long QT syndrome, and the Harveyras-1 gene.Science 1991;252: 704–706.
Meredith IT, Broughton A, Jennings GL, Esler MD. Evidence for a selective increase in resting cardiac sympathetic activity in some patients suffering sustained out of hospital ventricular arrhythmias.N Engl J Med 1991;325: 618–624.
La Rovere MT, Specchia G, Mortara A, Schwartz PJ. Baroreflex sensitivity, clinical correlates and cardiovascular mortality amongst patients with a first myocardial infarction.Circulation 1988;78: 816–824.
Akselrod S, Gordon D, Madwed JB, Snidman NC, Shannon DC, Cohen RJ. Hemodynamic regulation: investigation by spectral analysis.Am J Physiol 1985;249: H867-H875.
Guzzetti S, Piccaluga E, Casati R, Cerutti R, Lombardi F, Pagani M, Malliani A. Sympathetic predominance in essential hypertension: a study employing spectral analysis of heart rate variability.J Hypertens 1988;6: 711–717.
Farrell TG, Odemuyiwa O, Bashir Y, Cripps TR, Malik M, Ward DE, Camm AJ. Prognostic value of baroreflex sensitivity testing after acute myocardial infarction.Br Heart J 1992;67: 129–137.
Hasking G, Esler M, Jennings G, Button D, Johns J, Korner P. Norepinephrine spillover to plasma in congestive heart failure: evidence of increased overall and cardiorenal sympathetic nervous activity.Circulation 1986;73: 615–621.
Esler M, Jennings G, Lambert G. Measurement of overall and cardiac norepinephrine release into plasma during cognitive challenge.Psychoneuroendocrinology 1989;14: 477–481.
Wallin BG, Esler M, Dorward P, Eisenhofer G, Ferrier C, Westerman R, Jennings G. Simultaneous measurements of cardiac norepinephrine spillover and sympathetic outflow to skeletal muscle in humans.J Physiol (in press).
The Cardiac Arrhythmia Suppression Trial (CAST) Investigators. Preliminary report: effect of encainide and flecainide on mortality in a randomized trial of arrhythmia suppression after myocardial infarction.N Engl J Med 1989;321: 406–412.
Yusuf S, Peto R, Lewis J, Collins, Sleight P. Beta blockade during and after myocardial infarction: an overview of the randomized trials.Prog Cardiovasc Dis 1985;17: 335–371.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Esler, M. The autonomic nervous system and cardiac arrhythmias. Clinical Autonomic Research 2, 133–135 (1992). https://doi.org/10.1007/BF01819669
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01819669