Abstract
Two sibs with the acute neonatal form of glutaric aciduria type II (deficientin vivo activity of multiple acyl-CoA dehydrogenases) are described. In the second case diagnosis was made prenatally on the basis of reduced oxidation of palmitate by cultured amniotic fluid cells. With prompt intervention in the neonatal period and a carefully controlled diet later, this second case progressed well up to 4 months of age but died suddenly of cardiac failure, probably attributable to accumulation of fat. Neither patient showed any congenital morphological abnormality.
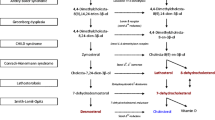
Cultured fibroblasts from the second case showed a marked defect in the oxidation of a range of substrates requiring acyl-CoA dehydrogenases for their catabolism, but residual activity for some substrates was quite high. Large quantities of sarcosine were excreted in urine, again suggesting that the mutation leaves some residual dehydrogenation activity. Butyryl-, octanoyl- and palmitoyl-CoA dehydrogenases were present in essentially normal quantities in postmortem liver.
Similar content being viewed by others
References
Blass, J. P., Avigan, J. and Uhlendorf, B. W. A defect in pyruvate decarboxylase in a child with an intermittent movement disorder.J. Clin. Invest. 49 (1970) 423–432
Blass, J. P., Schulman, J. D., Young, D. S. and Hom, E. An inherited defect affecting the tricarboxylic acid cycle in a patient with congenital lactic acidosis.J. Clin. Invest. 51 (1972) 1845–1851
Bohm, N., Uy, J., Kiessling, M. and Lehnert, W. Multiple acyl-CoA dehydrogenation deficiency (glutaric aciduria type II), congenital polycystic kidneys, and symmetric warty dysplasia of the cerebral cortex in two newborn brothers. II. Morphology and pathogenesis.Eur. J. Pediatr. 139 (1982) 60–65
Bradford, M. M. A rapid and sensitive method for the quantitation of protein using the principal of protein dye binding.Anal. Biochem. 72 (1978) 248–254
Coudé, F. X., Ogier, H., Charpentier, C., Thomassin, G., Checoury, A., Amédée-Manesme, O., Saudubray, J. M. and Frézal, J. Neonatal glutaric aciduria type II: an X-linked recessive inherited disorder.Hum. Genet. 59 (1981) 263–265
Goodman, S. I. and Frerman, F. E. Glutaric acidaemia type II (multiple acyl-CoA dehydrogenation deficiency).J. Inher. Metab. Dis. 7 Suppl. 1 (1984) 33–37
Goodman, S. I. and Markey, S. P.Diagnosis of Organic Acidemias by Gas Chromatography-Mass Spectrometry. Liss, New York, 1981, p. 72
Goodman, S. I., McCabe, E. R. B., Fennessey, P. V. and Mace, J. W. Multiple acyl Co-A dehydrogenase deficiency (glutaric aciduria type II) with transient hypersarcosinemia and sarcosinuria; possible inherited deficiency of an electron transfer flavoprotein.Pediatr. Res. 14 (1980) 12–17
Goodman, S. I., Stene, D. O., McCabe, E. R. B., Norenberg, M. D., Shikes, R. H., Stumpf, D. A. and Blackburn, G. K. Glutaric acidemia type II: clinical, biochemical and morphologic consideration.J. Pediatr. 100 (1982) 946–950
Goodman, S. I., Reale, M. and Berlow, S. Glutaric aciduria type II: A form with deleterious intrauterine effects.J. Pediatr. 102 (1983) 411–413
Gregersen, N., Kolvraa, S., Rasmussen, K., Christensen, E., Brandt, N. J., Ebbesen, F. and Hansen, F. H. Biochemical studies in a patient with defects in the metabolism of acyl-CoA and sarcosine: another possible case of glutaric aciduria type II.J. Inher. Metab. Dis. 3 (1980) 67–72
Lehnert, W., Wendel, U., Lindenmaier, S. and Bohm, N. Multiple acyl-CoA dehydrogenation deficiency (glutaric aciduria type II), congenital polycystic kidneys, and symmetric warty dysplasia of the cerebral cortex in two brothers. I. Clinical, metabolical and biochemical findings.Eur. J. Pediatr. 139 (1982) 56–59
Mitchell, G., Saudubray, J. M., Benoit, Y., Rocchiccioli, F., Charpentier, C., Ogier, H. and Boue, J. Antenatal diagnosis of glutaricaciduria type II.Lancet 1 (1983) 1099
Niederwieser, A., Steinmann, B., Exner, U., Neuheiser, F., Redweik, U., Wang, M., Rampini, S. and Wendel, U. Multiple acyl-CoA dehydrogenation deficiency (MADD) in a boy with nonketotic hypoglycemia, hepatomegaly, muscle hypotonia and cardiomyopathy. Detection ofN-isovaleryl-glutamic acid and its monoamide.Helv. Paediatr. Acta 38 (1983) 9–26
Olson, J. A. and Anfinsen, C. B. Kinetics and equilibrium studies on crystallinel-glutamate dehydrogenase.J. Biol. Chem. 202 (1953) 841–856
Przyrembel, H., Wendel, U., Becker, K., Bremer, H. J., Bruinvis, L., Ketting, D. and Wadman, S. K. Glutaric aciduria type II: report on a previously undescribed metabolic disorder.Clin. Chim. Acta 66 (1976) 227–239
Rhead, W., Mantagos, S. and Tanaka, K. Glutaric aciduria type II:In vitro studies on substrate oxidation, acyl-CoA dehydrogenases, and electron-transferring flavoprotein in cultured fibroblasts.Pediatr. Res. 14 (1980) 1339–1342
Saudubray, J-M., Coudé, F-X., Demaugre, F., Johnson, C., Gibson, K. M. and Nyhan, W. L. Oxidation of fatty acids in cultured fibroblasts: a model system for the detection and study of defects in oxidation.Pediatr. Res. 16 (1982) 877–881
Shaw, L. and Engel, P. C. Purification and properties of oxliver short-chain acyl-CoA dehydrogenase.Biochem. J. 218 (1984) 511–520
Sweetman, L., Nyhan, W. L., Trauner, D. A., Merritt, T. A. and Singh, M. Glutaric aciduria Type II.J. Pediatr. 96 (1980) 1020–1026
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bennett, M.J., Curnock, D.A., Engel, P.C. et al. Glutaric aciduria type II: Biochemical investigation and treatment of a child diagnosed prenatally. J Inherit Metab Dis 7, 57–61 (1984). https://doi.org/10.1007/BF01805802
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01805802