Summary
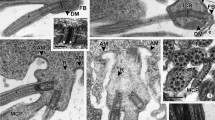
The multi flagellated sperm cells ofGinkgo biloba, differentiate within the pollen tube intra-ovularly in vivo. The sperm contain an anterior anti-clockwise spiral multi-layered structure (MLS) that is present in four of the grooves, and profiles vary in length from 15 μm in the center to < 5 μm in the outer grooves. The flagella show the typical 92 + 2 substructure. In about 5% of flagella, two of these 92 + 2 substructures occur within a single flagellar membrane, and up to 4 have been observed. The flagella are connected to the MLS through basal bodies, which have a long stellate transition zone terminating in the electron-opaque outer layer. This overlies the spline microtubules and an anterior fibrous lamellar strip. In transection, the MLS is terminated by paired fibrillar structures reminiscent of the blepharoplast.Ginkgo sperm cells contain numerous plastids and Golgi bodies, organelles that have not been reported from sperm of cycads.
Similar content being viewed by others
References
Bell P (1974) The origin of the multi-layered structure in the spermatozoid ofPteridium aquilinum. Cytobiologie 8: 203–212
—, Duckett JG, Myles D (1971) The occurrence of a multilayered structure in the motile spermatozoids ofPteridium aquumum. J Ultrastruct Res 34: 181–189
Carothers ZB, Kreitner GL (1967) Studies of spermatogenesis in theHepaticae. I. Ultrastructure of the Vierergruppe inMarchantia. J Cell Biol 33: 43–51
Gifford EM, Lin J (1974) Ultrastructure des cellules spermatogènes deGinkgo biloba, et particulièrement du blépharoplaste (mastigosome). C R Acad Sci (Paris) 278: 2513–2515
— — (1975) Light microscope and ultrastructural studies of the male gametophyte inGinkgo biloba: the spermatogenous cell. Am J Bot 62: 974–981
—, Larson S (1980) Developmental features of the spermatogenous cell inGinkgo biloba. Am J Bot 67: 119–124
Hirase S (1896) On the spermatozoid ofGinkgo biloba (in Japanese). Bot Mag (Tokyo) 10: 325–328
Marc J, Gunning BES (1987) Immunofluorescent localization of cytoskeletal tubulin and actin during spermatogenesis inPteridium aquilinum (L.) Kuhn. Protoplasma 134: 63–177
— —, Hardham AR, Perkin JL, Wick SM (1988) Monoclonal antibodies to surface and cytoskeletal components of the spermatozoid ofPteridium aquilinum. Protoplasma 142: 5–14
Moestrup ø (1982) Flagellar structure in algae: a review, with new observations particularly on theChrysophyceae, Phaeophyceae (Fucophyceae), Euglenophyceae, andReckertia. Phycologia 21: 427–528
Norstog K (1967) Fine structure of the spermatozoid ofZamia with special references to the flagellar apparatus. Am J Bot 54: 831–840
— (1974) Fine structure of the spermatozoid ofZamia: the Vierergruppe. Am J Bot 61: 449–456
— (1975) The motility of cycad spermatozoids in relation to structure and function. In: Duckett JG, Racey PA (eds) The biology of the male gamete. Academic Press, London, pp 135–142
— (1986) The blepharoplast ofZamia pumila L. Bot Gaz 147: 40–46
Paolillo DJ, Kreitner GL, Reighard JA (1968) Spermatogenesis inPolytrichum juniperinum I. The origin of the apical body and elongation of the nucleus. Planta 78: 226–247
Renzaglia KS, Carothers ZB, Duckett JG (1985) Comparitive ultrastructural studies of spermatogenesis in theMetzgeriales (Hepaticae). I. The blepharoplast ofPallavicinia lyellii. Am J Bot 72: 588–595
Tuohy M, McConchie CA, Knox RB, Szarski L, Arkin A (1987) Computer-assisted three-dimensional reconstruction technology in plant cell image analysis: applications of interactive computer graphics. J Microsc 147: 83–88
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Li, Y., Wang, F.H. & Knox, R.B. Ultrastructural analysis of the flagellar apparatus in sperm cells ofGinkgo biloba . Protoplasma 149, 57–63 (1989). https://doi.org/10.1007/BF01623983
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01623983