Conclusion
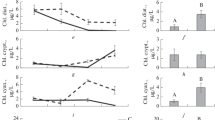
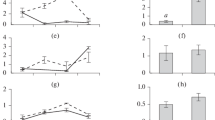
If we try to transfer these experimental results to phenomena which occur in the natural environment the phytoplanktonic biomasses, which have a high turnover rate during periods of growth, would show a very considerable facility to accumulate mercury, since the capacity to accumulate is similar for both compounds. But the first trophic transfers between these producers and the herbivores (Zooplankton for example) would show a marked inbalance in accumulation levels, in favour of methylmercury.
However, in this study we must take into consideration the very great diversity of biocoenoses and the interspecific differences which can manifest themselves with regard to processes of bioaccumulation and transfer. There is, moreover, continuous competition between the inert particles in suspension and the living organisms to absorb the mercury compounds which are in a free state in the aquatic environment. According to HAVLIK & al. (1979), certain phytoplanktonic species have a tendency to transform the bioaccumulated organic compounds: in this case the eff icacity of the transfer processes would depend on the extent to which the phenomenon occurs and the speed with which it begins.
With the results we have so far collected it is possible to suppose that the large quantities of the methylated form of mercury to be found in the terminal aquatic consumers — especially carnivorous fish II or III — can be accounted for by the mechanisms of bioaccumulation and transfer of the mercury compounds at the lower end of the trophic chains.
At this stage in our research we are interested in knowing how transfers between the primary consumer level and the secondary consumer lever occur. Is it the same process as that observed between the first two links in the chain, or is it slightly modified ? By using our experimental trophic chain —Chlorella vulgaris→Daphnia magna→Salmo gairdneri(newlyhatched) we should be able to answer this question, provided that it proves possible to quantify the transfer and bioaccumulation processes according to the two contamination routes.
Similar content being viewed by others
References
BOUDOU, A., A. DELARCHE, F. RIBEYRE and R. MARTY: Bulletin Environmental Contamination Toxicology22, 813 (1979).
DELARCHE, A., and F. RIBEYRE: Thèse n∘ 1436, Université de Bordeaux I, (1978).
FAREY, B.J., L.A. NELSON and M.G. ROLPH: Analyst103, 656 (1978).
FREAR, E.H., and J.E. BOYD: Journal Economic Entomology60(5), 1223 (1968).
HAVLIK, B., J. STARY and J. PRASILOVA: Acta Hydrochimica Hydrobiologica7(4), 401 (1979).
HUCKABEE, J.W., J.W. LEWOOD and S.G. Hilderrand in The biogeochemistry of mercury in the environment, Nriagu J.O., Elsevier North Holland (1979).
KNAUER, G.A., and J.H. MARTIN: Limnology and Oceanography17(6), 868 (1972).
RIBEYRE, F., A. BOUDOU and A. DELARCHE: Ecotoxicology and Environmental Safety3, 411 (1979).
RIBEYRE, F. and A. BOUDOU: Aquatic Toxicology (in press).
SELL, J.L., and K.L. DAVISON: Journal Agricultural Food Chemistry23(4), 803 (1975).
THELLEN, C., G. JOUBERT and R. Van COILLIE: Canadian Report Fish Aquatic Science (in press).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Boudou, A., Ribeyre, F. Comparative study of the trophic transfer of two mercury compounds—HgCl2 and CH3HgCl—BetweenChlorella vulgaris andDaphnia magna. Influence of temperature. Bull. Environ. Contam. Toxicol. 27, 624–629 (1981). https://doi.org/10.1007/BF01611073
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01611073